Previous studies have shown positive results of photobiomodulation (PBM) for improving performance and accelerating post-exercise recovery. However, the effects of PBM in healthy individuals who underwent a neuromuscular adaptation training remain unclear.
ObjectiveTo investigate the effects of PBM during a training program combining sprints and explosive squats exercises on clinical, functional, and systemic outcomes in trained healthy individuals compared to a placebo intervention and a control.
MethodsWe conducted a randomized placebo-controlled trial. Healthy males were randomly assigned to three groups: active PBM (30 J per site), placebo, or control (passive recovery). The participants performed a six-week (12 sessions) of a training program consisting of a combination of sprints and squats with recovery applied between sprints and squats. To prevent the influence of the primary neuromuscular adaptation to exercise on the results, all participants had to participate in a period of six weeks of exercise training program. Functional, clinical, and psychological outcomes and vascular endothelial growth factor (VEGF) were assessed at baseline and after six weeks. Results are expressed as mean difference (MD) and 95% confidence intervals (CI).
ResultsThirty-nine healthy male volunteers (aged 18–30 years; body mass index 23.9 ± 3 kg/m²) were recruited. There was no significant time by group interaction, and no significant effect of group, but there was a significant effect of time for maximal voluntary isometric contraction (primary outcome) (MD=22 Nm/kg; 95%CI: 3.9, 40) and for squat jump (MD=1.6 cm; 95CI%: 0.7, 2.5). There was no significant interaction (time*group), time, or group effect for the other outcomes.
ConclusionThe addition of PBM to a combined training performed for six weeks in previously trained individuals did not result in additional benefits compared to placebo or no additional intervention.
Photobiomodulation (PBM) therapy using low-level laser therapy (LLLT) and/or light emitting diodes therapy (LEDT) has been the focus of recent research. The PBM mechanism of action consists of the interaction of light with chromophores which influences cellular activity through the inhibition or stimulation of chemical and biological functions,1 such as synthesis of adenosine triphosphate (ATP),2 modulation of gene expression,3 increases in local microcirculation,4 and upregulated angiogenesis with vascular endothelial growth factor (VEGF) as mediator.5,6 This intervention has been associated with two main physiological effects, ergogenic and protective.7 As a consequence, PBM has been used as a prevention and rehabilitation intervention, including for post-exercise recovery, due to its positive results in a range of outcomes, as discussed in recent systematic reviews.8,9 However, the magnitude of the effects reported may be influenced by the treatment parameters used, such as the wavelength, energy, density, and power, as well as the type of tissue damage and time of application.10
A systematic review published by our research group suggests that PBM can have a different effect between general and localized exercises.9 Recent recommendations for the use of PBM suggest that there may be a differentiation on the exact moment of irradiation to obtain acute or chronic effects.11 These recommendations are in accordance with the consensus statement by the International Olympics Committee which highlighted the potential role of this intervention to improve acute muscle recovery.11,12
The use of PBM to promote chronic training effects began to be investigated in the last decade in humans.13–18 Variations with regards to the types of training and the best moment to apply the intervention have been reported. Regarding the type of training, studies have investigated the effects of PBM mainly on strength training13,17 and endurance.14,16 Positive effects have been reported when PBM was applied before the exercise, for outcomes such as time until exhaustion and number of repetitions,19 maximal voluntary contraction,20 creatine kinase (CK) and lactate,21 and when PBM was applied after the exercise, for outcomes such as peak torque and 1 repetition maximum (RM) test.22
Taken together, these preliminary findings suggest that the ergogenic and protective effects may be enhanced when the PBM is applied during a training session that combines strength and endurance exercises.23 In addition, to investigate the real effect of PBM we would argue that this intervention should be administered after the primary neuromuscular adaptation to exercise has already occurred. Therefore, the objective of this randomized placebo-controlled trial was to investigate the effects of PBM, administered during a training program consisting of a combination of sprints and explosive squats exercises, on clinical, functional, and systemic outcomes in trained healthy individuals compared to a placebo intervention and a control. The study hypothesis was that the use of a PBM applied during an exercise training program would accelerate the recovery process and improve performance.
MethodsTrial designThis study is a three-arm randomized placebo-controlled trial. This study was approved by the Human Research Ethics Committee of the Universidade Estadual Paulista (Protocol number: 1.389.046/2016) and registered at ClinicalTrials.gov (ID: NCT02918916). Prior to the start of the trial, participants received oral and written instructions regarding the procedures and objectives of the study and signed an informed consent form agreeing to participate in the study.
ParticipantsThe study was conducted at Universidade Estadual Paulista. Participants were recruited from the Laboratory of Physical Therapy in Sports database via online media, telephone, or text message. Participants included in this study met the following inclusion criteria: male aged between 18 and 30 years old and considered healthy (by self-report). Individuals who reported history of muscle injury in the lower limbs or spine in the previous six months were excluded from the study. Participants were instructed to maintain their daily diet routine and to continue with their daily exercise routine, without change in training volume or intensity. All participants were instructed to ingest 30 g of carbohydrate-based gel supplement followed by 180 ml of water 20 min before the baseline assessment or training to standardize the energy reserve.
Sample sizeThe sample size calculation was based on data from a previous study24 that used the same PBM device for a three-arm parallel study to detect a 10% difference between groups on maximal voluntary isometric contraction (MVIC), with standard deviation (SD) of 28.71 Nm, with significance set at 0.05 and power at 0.8. A total sample of 11 per group was required.
Randomization procedureA stratified randomized procedure was performed based on the concept of responders and non-responders to adaptation capacity.25 The training adaptation capacity was recorded before and after the first six-week of the exercise training program. The adaptation capacity was calculated using the equation AC=0.5FP+0.5CP, where AC is adaptation capacity, FP is the functional parameter (i.e. strength test) and CP is the clinical parameter (i.e. perception of recovery). Participants were first stratified into three strata (tertiles) based on their adaptation capacity, then participants from each stratum were allocated into one of the three groups by block randomization. To ensure allocation concealment, randomization procedure was performed by an independent researcher who was not involved in the recruitment of participants and outcome assessment. The complete randomization process was described in the published protocol.23
InterventionsTo prevent the influence of the primary neuromuscular adaptation to exercise on the results, all participants had to participate in a period of six weeks of exercise training. This training period aimed to induce neuromuscular adaptation and equalize the level of physical activity among the participants. After this period, the participants were randomly allocated, according to their adaptation capacity, into one of the three groups that comprise the study: active PBM group (AG), placebo PBM group (PG) and control group (CG). Participants in each of the groups performed another six-week of exercise training with the adjusted loads and received the intervention they were allocated to between sprint and squats training.
Training programThe training program was adapted from an individualized combined training described by Marques et al.26 The training consisted of a combination of sprints and explosive squats performed twice per week for six-weeks (48 h between sessions). A detailed description of the training is presented in Table 1.
Training program.
| Week | Sessions | Sprint Training | Squat Training | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Warm-up* | Intensity† | Sets x Repetitions | Rest between sets | Warm-up* | Intensity†∆ | Sets x Repetitions | Rest between sets | |||
| 1 | 1 | 3 × 20m | Maximum | 2 × 15m | 120s | 10 minutes between training | 1 × 8+1 × 8 | 80% | 1 × 8 | 120s |
| 2 | 3 × 20m | 2 × 15m | 120s | 1 × 8+1 × 8 | 80% | 2 × 8 | 120s | |||
| 2 | 3 | 3 × 25m | 2 × 20m | 120s | 1 × 8+1 × 8 | 85% | 1 × 8 | 120s | ||
| 4 | 3 × 25m | 2 × 20m | 120s | 1 × 8+1 × 8 | 85% | 2 × 6 | 120s | |||
| 3 | 5 | 3 × 30m | 2 × 25m | 150s | 1 × 8+1 × 8 | 90% | 1 × 6 | 120s | ||
| 6 | 3 × 30m | 2 × 25m | 150s | 1 × 8+1 × 8 | 90% | 2 × 8 | 120s | |||
| 4 | 7 | 3 × 35m | 2 × 30m | 180s | 1 × 8+1 × 6 | 95% | 1 × 6 | 180s | ||
| 8 | 3 × 35m | 2 × 30m | 180s | 1 × 8+1 × 6 | 95% | 2 × 5 | 180s | |||
| 5 | 9 | 3 × 25m | 2 × 20m | 150s | 1 × 8+1 × 6 | 100% | 2 × 6 | 180s | ||
| 10 | 3 × 25m | 2 × 20m | 150s | 1 × 8+1 × 6 | 100% | 2 × 5 | 180s | |||
| 6 | 11 | 3 × 30m | 1 × 25m | 180s | 1 × 8+1 × 6 | 90% | 2 × 6 | 120s | ||
| 12 | 3 × 30m | 1 × 25m | 180s | 1 × 8+1 × 6 | 80% | 1 × 8 | 120s | |||
Warm-up: sprints in which speed is gradually increasing until the participant reaches the maximum speed; squats with lighter loads to those in training (40–60% of the perceived maximum).
Recovery strategies were applied between sprint and squats training due to the preliminary hypothesis of possible association of ergogenic and protective effects of PBM in the same session.
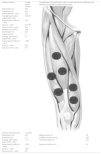
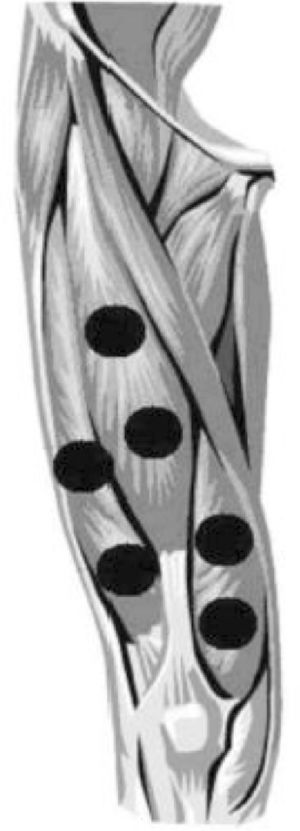
Active PBM was applied using a MR4 LaserShower 50 4D emitter (Multi Radiance Medical, USA), to six sites of the quadriceps (two centrally – rectus femoris and vastus intermedius; two laterally – vastus lateralis; two medially – vastus medialis).24 The decision to apply the intervention at these sites was made because this muscle group is most directly involved during squats and sprints. The dosage (30 J per site; 180 J per muscle) was based on findings of Antonialli et al.24 More details are provided in Table 2.
Photobiomodulation parameters.
The same procedures were applied to the placebo group; however, the PBM probe was disabled. The therapist responsible for the application of active and placebo PBM was not aware of the equipment settings which was programmed by an independent researcher.
The control group participants performed passive recovery (remained seated) supervised by an independent researcher while the other groups were receiving the recovery strategies. Participants from all groups remained seated to receive the recovery strategies.
OutcomesThe MVIC was considered the primary outcome. Secondary outcomes were other functional and clinical outcomes and VEGF, as described below. The same assessor was responsible for the outcome assessment throughout the study. Outcome assessment was completed over two sessions separated by 24 h. The first session consisted of sprint and squat jump tests. In the second session, strength and power tests and MVIC measurements were performed. An interval of 15 min between tests was used to limit effects of fatigue on subsequent test. This rest is based on time to recovery of each test described in the literature.27–31
Primary outcomeMaximal voluntary isometric contraction (MVIC): Participants were positioned with the dominant leg on the isokinetic dynamometer (Biodex System 4 Pro, New York, USA). Prior to the MVIC, a warm-up consisting of 10 repetitions of knee flexion-extension at 180°/s throughout the range of motion was performed. The MVIC was determined by the highest maximal isometric torque over three contractions of five seconds at 60° of knee flexion. The three contractions were separated by a 2-minute rest interval.20
Secondary outcomesStrength and power tests: Tests were performed using a guided bar.26 Participants were required to perform the squat with continuous movement until their thighs reached the horizontal plane, and immediately to perform the opposite movement at maximum speed without losing feet contact with the ground. For the strength test, the warm-up consisted of 10 repetitions at 40–60% of perceived maximum. A progression from warm-up load was performed with increment of 10 kg. Two minutes rest interval between sets were provided. The last acceptable repetition with the highest possible load was considered as 1-RM. For the power test, the warm-up consisted of five repetitions with the bar. To determine the load moved at 1 m/s the tests were conducted gradually with increments of 10 kg for each set and two attempts for each load. An interval of 3 min was given between attempts. The highest measure with each load was recorded. Information about the distance held eccentrically and concentric speed of each repetition were recorded by a linear velocity transducer (T-Force System Ergotech, Murcia, Spain) and analyzed by its specific software (Isocontrol Dinámico, 3.6, Spain).
Squat jump test: Participants performed three squat jumps on a platform without countermovements (Multisprint, Hidrofit, Brazil). A rest interval of 30 s was considered between jumps. The best trial was used for data analysis.32
Sprint test: Participants performed three maximum sprints exceeding 30 m with rest intervals of three minutes. Performance times at 10, 20, and 30 m were recorded by phothocells (Multisprint, Hidrofit, Brazil). The average of the two best measures was used for data analysis.26
Muscle soreness: Values of muscle soreness in the lower limbs were obtained by a 0–10 points visual analog scale (VAS),20 with ‘zero’ corresponding to absence of soreness and ‘10’ to the maximum soreness a participant felt could be tolerated.33
Pain threshold: Pressure algometry was applied at five specific sites of the quadriceps of the dominant leg.34 Participants were instructed to indicate when the pressure sensation became discomfort. The pressure did not exceed 2.55 kgf.35
Psychological questionnaire: Participants filled out a questionnaire about readiness for exercise, fatigue, vigor, and sleepiness. The information about pain was excluded due to use of a pain VAS.36
Vascular endothelial growth factor (VEGF): 10 ml of blood was collected 24 h before the training and 24 h after the last training session. The plasma from this sample was stored at −80 °C for later analysis. The plasma concentration of VEGF was analyzed using the ELISA method (enzyme-linked immunosorbent assays) following the manufacturer's instructions (R&D Systems, Minneapolis, MN, USA).37
Statistical methodsThe statistical analysis was conducted using the software SPSS (version 22; SPSS Inc, Chicago, Illinois, USA). Overall, data were normally distributed as tested by Shapiro-Wilk test and visually confirmed by Q-QPlots. Goodness-of-fit was tested by Akaike's Information Criterion (AIC) and therefore the General Mixed Model (GMM) with time and group as fixed factors, subjects as random factor and AR(1) matrix was performed for all outcomes. All tests were performed assuming a significance level of p < 0.05, including Bonferroni post-hoc when main effects were found. Results are expressed as mean difference estimates and standard deviation (SD) with 95% confidence intervals (CI) for group comparisons. Participants who dropped out of the study were excluded from the analysis.
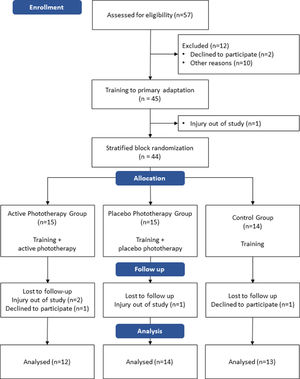
ResultsThirty-nine healthy (self-report) male volunteers (aged 18–30 years; height 176.9 ± 6.2 cm; weight 75.3 ± 12 kg; body mass index 23.9 ± 3 kg/m²) recruited between January and March 2017, completed the study. The flowchart of participants is presented in Fig. 1.
Table 3 shows the results for functional, clinical, and psychological outcomes. There was no significant time by group interaction, and no significant effect of group. However, there was a significant time effect for MVIC (P = 0.018) and squat jump (12.60, P = 0.001). Bonferroni post-hoc corrections showed that, after six weeks, there was an increase in MVIC (MD=33.5 Nm/kg; 95%CI= 2.4, 64.7) and squat jump (MD=1.9 cm; 95%CI=0.3, 3.5) regardless of the intervention group. There were no significant main effects for the remaining outcomes (Table 3; Fig. 2).
Primary and secondary outcomes.
| Within group differences: mean ± SD (95% CI) | Between group differences: mean ± SD (95%CI) | |||||
|---|---|---|---|---|---|---|
| Active group (n = 12) Mean ± SD (95%CI) | Placebo group (n = 14) Mean ± SD (95%CI) | Control group (n = 13) Mean ± SD (95%CI) | Active - Placebo | Active - Control | Placebo - Control | |
| Primary outcome | ||||||
| MVIC (peak torque/body weight) | ||||||
| Baseline | 318.0 ± 59.2 | 316.8 ± 59.5 | 312.5 ± 59.2 | -2.5 ± 21.8(-45.2, 40.2) | -18,5 ± 22.6(-61.9, 24.9) | -16.0 ± 21.3(-57.8, 25.8) |
| At 6 wks | 333.0 ± 75.0 | 334.3 ± 75.0 | 346.1 ± 75.0 | |||
| Change baseline → 6 wks | 15.0 ± 55.4(-17.4, 47.4) | 17.5 ± 55.4(-12.5, 47.5) | 33.5 ± 55.3(2.4, 64.7) | |||
| Secondary outcomes (functional) | ||||||
| 1 RM (kg) | ||||||
| Baseline | 69.0 ± 14.9 | 64.3 ± 16.4 | 71.8 ± 14.9 | 3.7 ± 3.4(-3.0, 10.4) | 1.7 ± 3.5(-5.1, 8.5) | -2.0 ± 3.4(-8.6, 4.6) |
| At 6 wks | 71.9 ± 16.4 | 63.5 ± 14.9 | 73.00 ± 16.4 | |||
| Change baseline → 6 wks | 2.9 ± 8.7(-2.2, 8.0) | -0.8 ± 8.7(-5.5, 3.9) | 1.2 ± 8.7(-3.7, 6.1) | |||
| Load lifted at 1m/s | ||||||
| Baseline | 32.2 ± 13.5 | 30.3 ± 13.5 | 30.2 ± 13.5 | -2.1 ± 5.1(-12.1, 7.9) | -7.7 ± 5.2(-17.8, 2.5) | -5.6 ± 4.9(-15.3, 4.2) |
| At 6 wks | 29.1 ± 14.6 | 29.3 ± 14.6 | 34.7 ± 14.6 | |||
| Change baseline → 6 wks | -3.1 ± 12.9(-10.75, 4.5) | -1.0 ± 12.9(-8.0, 6.0) | 4.6 ± 12.9(-2.7, 11.9) | |||
| Squat jump (cm) | ||||||
| Baseline | 32.0 ± 5.5 | 29.5 ± 5.5 | 30.8 ± 5.5 | -2.7 ± 1.1(-4.84, -0.51) | -1.8 ± 1.1(-3.9, 0.4) | 0.9 ± 1.1(-1.2, 2.9) |
| At 6 wks | 32.1 ± 5.3 | 32.3 ± 5.3 | 32.7 ± 5.3 | |||
| Change baseline → 6 wks | 0.1 ± 2.8(-1.5, 1.8) | 2.8 ± 2.8(1.3, 4.3) | 1.9 ± 2.8(0.3, 3.5) | |||
| Sprint 10 m (s) | ||||||
| Baseline | 1.7 ± 0.1 | 1.7 ± 0.1 | 1.7 ± 0.1 | -0.0 ± 0.0(-0.0, 0.0) | -0.0 ± 0.0(-0.0, 0.0) | -0.0 ± 0.0(-0.0, 0.0) |
| At 6 wks | 1.7 ± 0.1 | 1.8 ± 0.1 | 1.7 ± 0.1 | |||
| Change baseline → 6 wks | -0.0 ± 0.1(-0.0, 0.0) | -0.0 ± 0.0(-0.0, 0.0) | 0.0 ± 0.1(-0.0, 0.0) | |||
| Sprint 20 m (s) | ||||||
| Baseline | 3.1 ± 0.2 | 3.2 ± 0.2 | 3.1 ± 0.2 | -0.0 ± 0.0(-0.1, 0.1) | 0.0 ± 0.0(-0.1, 0.1) | 0.0 ± 0.0(-0.1, 0.1) |
| At 6 wks | 3.1 ± 0.2 | 3.2 ± 0.2 | 3.1 ± 0.2 | |||
| Change baseline → 6 wks | 0.0 ± 0.1(-0.1, 0.1) | 0.0 ± 0.1(-0.1, 0.1) | -0.0 ± 0.1(-0.1, 0.1) | |||
| Sprint 30 m (s) | ||||||
| Baseline | 4.5 ± 0.3 | 4.5 ± 0.3 | 4.4 ± 0.3 | -0.0 ± 0.0(-0.1, -0.0) | -0.0 ± 0.0(-0.1, 0.1) | 0.0 ± 0.0(-0.1, 0.1) |
| At 6 wks | 4.4 ± 0.3 | 4.5 ± 0.3 | 4.4 ± 0.3 | |||
| Change baseline → 6 wks | -0.0 ± 0.1(-0.1, 0.0) | 0.0 ± 0.1(-0.1, 0.1) | -0.0 ± 0.1(-0.1, 0.1) | |||
| Secondary outcomes (clinical) | ||||||
| Muscle soreness | ||||||
| Baseline | 0.3 ± 0.6 | 0.1 ± 0.6 | 0.2 ± 0.6 | 0.1 ± 0.4(-0.7, 0.9) | -0.1 ± 0.4(-0.9, 0.7) | -0.2 ± 0.4(-0.9, 0.6) |
| At 6 wks | 0.4 ± 0.9 | 0.1 ± 0.9 | 0.4 ± 0.9 | |||
| Change baseline → 6 wks | 0.2 ± 1.0(-0.4, 0.8) | 0.1 ± 1.0(-0.5, 0.6) | 0.2 ± 1.0(-0.3, 0.8) | |||
| Pain threshold (kgf) | ||||||
| Baseline | 2.4 ± 0.4 | 2.3 ± 0.4 | 2.4 ± 0.4 | -0.0 ± 0.2(-0.4, 0.3) | 0.1 ± 0.2(-0.3, 0.4) | 0.1 ± 0.2(-0.2, 0.4) |
| At 6 wks | 2.4 ± 0.3 | 2.4 ± 0.3 | 2.4 ± 0.3 | |||
| Change baseline → 6 wks | 0.0 ± 0.4(-0.3, 0.2) | 0.1 ± 0.4(-0.2, 0.3) | -0.0 ± 0.4(-0.3, 0.2) | |||
| Physically ready* | ||||||
| Baseline | 8.5 ± 1.7 | 7.9 ± 1.7 | 8.4 ± 1.7 | 0.2 ± 0.9(-1.6, 1.9) | 0.2 ± 0.9(-1.6, 1.9) | 0.0 ± 0.8(-1.6, 1.7) |
| At 6 wks | 8.6 ± 1.7 | 7.8 ± 1.7 | 8.3 ± 1.7 | |||
| Change baseline → 6 wks | 0.1 ± 1.2(-1.2, 1.4) | -0.1 ± 2.2(-1.3, 1.2) | -0.1 ± 2.2(-1.3, 1.2) | |||
| Mentally ready* | ||||||
| Baseline | 9.1 ± 2.1 | 7.7 ± 2.2 | 8.4 ± 2.2 | -0.5 ± 0.6(-1.7, 0.7) | -0.0 ± 0.6(-1.3, 1.2) | 0.5 ± 0.6(-0.7, 1.7) |
| At 6 wks | 9.4 ± 1.1 | 8.5 ± 1.1 | 8.7 ± 1.1 | |||
| Change baseline → 6 wks | 0.3 ± 1.6(-0.6, 1.2) | 0.8 ± 1.6(-0.1, 1.7) | 0.3 ± 1.6(-0.6, 1.2) | |||
| Fatigue* | ||||||
| Baseline | 0.8 ± 1.6 | 2.3 ± 1.6 | 1.9 ± 1.6 | 0.8 ± 0.6(-0.3, 1.9) | 0.1 ± 0.6(-0.9, 1.2) | -0.7 ± 0.5(-1.7, 0.4) |
| At 6 wks | 1.0 ± 1.7 | 1.8 ± 1.7 | 2.1 ± 1.7 | |||
| Change baseline → 6 wks | 0.3 ± 1.4(-0.6, 1.1) | -0.5 ± 1.4(-1.3, 0.3) | 0.1 ± 1.4(-0.7, 0.9) | |||
| Vigorous*⁎ | ||||||
| Baseline | 8.5 ± 1.7 | 7.1 ± 1.7 | 7.7 ± 1.7 | -1.6 ± 0.7(-2.9, -0.3) | -0.4 ± 0.7(-1.7, 0.9) | 1.2 ± 0.7(-0.1, 2.5) |
| At 6 wks | 8.2 ± 1.6 | 8.4± 1.5 | 7.8 ± 1.6 | |||
| Change baseline → 6 wks | -0.3 ± 1.7(-1.3, 0.7) | 1.3 ± 1.7(0.4, 2.2) | 0.1 ± 1.7(-0.9, 1.1) | |||
| Sleepy* | ||||||
| Baseline | 2.0 ± 1.2 | 1.9 ± 1.2 | 2.6 ± 2.2 | 0.9 ± 0.6(-0.3, 2.2) | 1.3 ± 0.7(-0.0, 2.5) | 0.3 ± 0.6(-0.9, 1.5) |
Abbreviations: MVIC, maximal voluntary isometric contraction); SD, standard deviation; CI, confidence interval.
This study aimed to assess the effects of PBM during a six-week training program, combining sprints and explosive squats exercises, on functional, clinical, and systemic outcomes in trained individuals compared to a placebo intervention and a control (i.e. passive recovery). Our findings showed that PBM was not superior to placebo and control in any of the investigated outcomes.
A previous meta-analysis from our research group9 identified that the effects of PBM on the concentration of CK depend on the type of stress, showing greater effects after localized exercises. Therefore, we understand that PBM may promote better results when exercise causes greater muscular damage indicated, for example, by higher concentrations of CK. Accordingly, we chose an exercise training program that combines sprints and explosive squats because it has been proposed to optimize performance adaptations, suggesting higher gains than single-element programs.38 However, we found only small variations in clinical and psychological outcomes throughout the groups suggesting that training program was able to promote positive adaptations such as improved MVIC and squat jump performance without causing major stress. It is suggested that after the process of primary adaptation to exercise, individuals may experience less stiffness, pain, and less muscle damage,39 which may have occurred after the initial six weeks of training. Therefore, because the combined training did not yield significant pain and loss of function over the training period the PBM effects might have been attenuated.
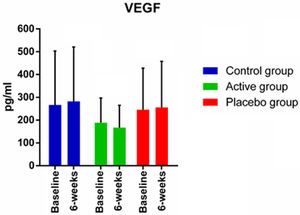
Although some authors believe that PBM can act systemically, no changes were observed in the VEGF variable when comparing to the placebo and control groups. The VEGF is considered a systemic angiogenic cytokine and its production can occur in peripheral and central areas.40 Its release mechanisms are mainly related to the hemodynamic stimulus and can be influenced by exercise intensity.41,42 Skriver et al.37 point out that changes in the concentration of this marker in humans after exercise are not yet fully elucidated, with divergent results.
In a systematic review involving the effects of exercise on VEGF, Vital et al.43 discusses that training frequencies less than three times a week may not be enough to promote increased VEGF production in the elderly. Similarly, as observed in the present study, regarding a healthy and trained population adapted to exercise, the frequency of twice a week does not seem to be sufficient to change VEGF production, as can be seen in the control group. Furthermore, the results also suggest that PBM was not able to promote additional effects, with no difference between the active PBM group and the other groups.
Vieira et al.44 identified a 31% increase in VEGF concentration in the group that performed strength training associated with PBM. However, the authors stress that these effects come from preliminary results, which does not allow to identify definitive conclusions.44 Thus, from the findings of this study, it is suggested that the addition of PBM applied bilaterally twice a week to a combined training of six weeks does not cause any systemic and functional benefits.
The study presents high methodological quality with a noteworthy design. The stratified block randomization allowed a better distribution of responders and non-responders into groups. This type of control has been considered important and innovative when considering inter and intra-variability responses. The application protocol performed after control of the primary adaptation process resulting from the exercise may be able to reflect the real effect of the technique, which can be considered a strength of the study. To our knowledge this is the first study to investigate the effect of PBM during a combined training. The study is also the pioneer in considering participants previously adapted to the stimulus provided by the specific type of training. Thus, the control of the primary adaptation process (trained subjects) may be the justification for which benefits of PBM were not observed.
The PBM was applied on strategic sites according to greater muscle demand. Furthermore, we understand that the application method with simultaneous probes can be considered a limitation for clinical practice. Thus, future investigations should consider the application of PBM to other muscle groups used during this type of training, in addition to those applied in this study (i.e. hamstring, gastrocnemius), as well as the use of equipment that involves all the musculature. Such options should be investigated to verify whether the absence of systemic results and muscle function are confirmed even with the use of other methods of application.
ConclusionThe results of this study show that the addition of PBM to a combination of squat and sprint training performed for six weeks in previously trained individuals did not result in additional benefits compared to the same training program supplemented by a placebo intervention or no additional intervention (control).
This work was supported by the São Paulo Research Foundation – FAPESP under Grant [2015/25220-9; 2015/25219-0; 2017/24900-1]; and National Counsel of Technological and Scientific Development – CNPq.