The evidence of the influence of Kinesio Taping® in changing electromyographic signal intensity of the lumbar musculature in patients with chronic non-specific low back pain (LBP) is very sparse.
ObjectivesTo evaluate if Kinesio Taping® changes the electromyographic signal intensity of the longissimus and iliocostalis muscles in patients with chronic non-specific LBP.
MethodsProspectively registered, three-arm randomized controlled trial with a blinded assessor. Patients were randomly allocated to the following interventions: 1) Kinesio Taping® Group (n=21), where patients received the tape according to the manufacturer's manual; 2) Placebo Group (i.e. normal surgical tape) (n=21); and 3) Non-treatment control Group (n=21). Assessments were performed at baseline, immediately after, and 30min after the intervention. The primary outcome was muscle activity of the iliocostalis and longissimus muscles as measured by surface electromyography. The secondary outcome was pain intensity (measured with a 0–10 Numerical Rating Scale). The effects of treatment were calculated using linear mixed models.
ResultsA total of 63 patients were recruited. Follow up rate was high (98.4%). Patients were mostly women with moderate levels of pain and disability. Kinesio Taping® was better than the control and placebo groups in only 4 of 96 statistical comparisons, likely reflective of type I error due to multiple comparisons. No statistically significant differences were identified for the immediate reduction in pain intensity between groups.
ConclusionKinesio Taping® did not change the electromyographic signal intensity of the longissimus and iliocostalis muscles or reduce pain intensity in patients with chronic low back pain.
Clinicaltrials.gov: NCT02759757 (https://clinicaltrials.gov/ct2/show/NCT02759757)
Low back pain (LBP) is one of the most prevalent public health issues worldwide.1 According to the most recent Global Burden of Disease Study, LBP is the leading cause of years lived with disability.2,3 Episodes of acute LBP usually have a good prognosis, with significant improvement in the first few weeks.4 After this time, however, more than 40% of patients are likely to develop chronic LBP (pain that persists for more than 12 weeks).5 Studies have shown that the use of health services due to LBP has increased in recent years,6 and patients with chronic LBP tend to seek more treatment than patients with acute LBP.7,8
Non-pharmacological interventions such as exercise, manual therapy, psychological therapies, and multidisciplinary biopsychosocial rehabilitation9 are the first options in the treatment of patients with chronic LBP.9–11 The latest guidelines published for the treatment of LBP have recommended that medication should only be used in patients who have not improved with non-pharmacological treatment due to the potential adverse effects and limited effectiveness of drugs.9,12
Kinesio Taping (KT)® has become very popular, especially among athletes,13 and has also been proposed for the treatment of patients with LBP.14,15 KT is an intervention consisting of the application of a porous, adhesive elastic bandage, without chemicals or drugs.16 KT can extend up to 140% of its original length, allowing good range of motion when compared to other types of tape.16,17 The creators of the technique claim that KT reduces pain intensity and abnormal muscle tension, change joint position and movement perception, and reduces muscle spasms by reducing muscle activation, among other mechanisms of action.17,18 Patients with chronic LBP typically experience moderate levels of pain and disability. In addition, superficial trunk muscles (such as the longissimus and iliocostalis muscles) are usually over-activated in response to pain stimulus.19,20,21
Although the creators of the technique propose that KT reduces muscle activation (ie, electromyographic (EMG) signal intensity), only three studies,15,22,23 to date, have investigated this possible mechanism. Because these trials presented methodological limitations related to study design, small sample size, and inadequate description of EMG parameters, further investigation is warranted. Therefore, we conducted a clinical trial aimed to determine the effects of Kinesio Taping® on the EMG signal intensity of the lumbar musculature of patients with chronic LBP.
MethodsDesignA three-arm, randomized controlled trial with a blinded assessor was conducted.
Ethics and clinical trial registrationThe project was approved by the Research Ethics Committee of Universidade Cidade de São Paulo (UNICID), São Paulo, Brazil (#50548115.8.0000.0064) and was prospectively registered on Clinicaltrials.gov (NCT02759757). All eligible participants were informed about the objectives of the study and were asked to complete an informed consent form. The study was conducted at the Physical Therapy Clinic of the Universidade Cidade de São Paulo.
Study populationThe present study included participants of both sexes, aged between 18 and 60 years, presenting with chronic non-specific LBP (pain or discomfort with duration of at least three months between the costal margins and the inferior gluteal folds, with or without referred pain to the lower limbs). Patients who had previously used KT for their LBP and/or any other adhesive tape and patients with any contraindication to their use were excluded from the study. Other exclusion criteria were: serious spinal pathologies (fracture, tumor, inflammatory or infectious disease), previous spinal surgery, patients with nerve root compromise, decompensated cardiorespiratory and metabolic diseases, pregnancy, and previous use of KT for any condition.
ProcedureInitially, an individual physical examination was conducted,6 including collection of sociodemographic (such as age, gender, marital status, and education levels) and anthropometric data (such as weight, height, and body mass index) and assessment of pain intensity using the 11-point Numeric Pain Rating Scale (NPRS).24 Disability was assessed with the Roland-Morris Disability Questionnaire (RMDQ)25 scored from 0 (no disability) to 24 (high disability).
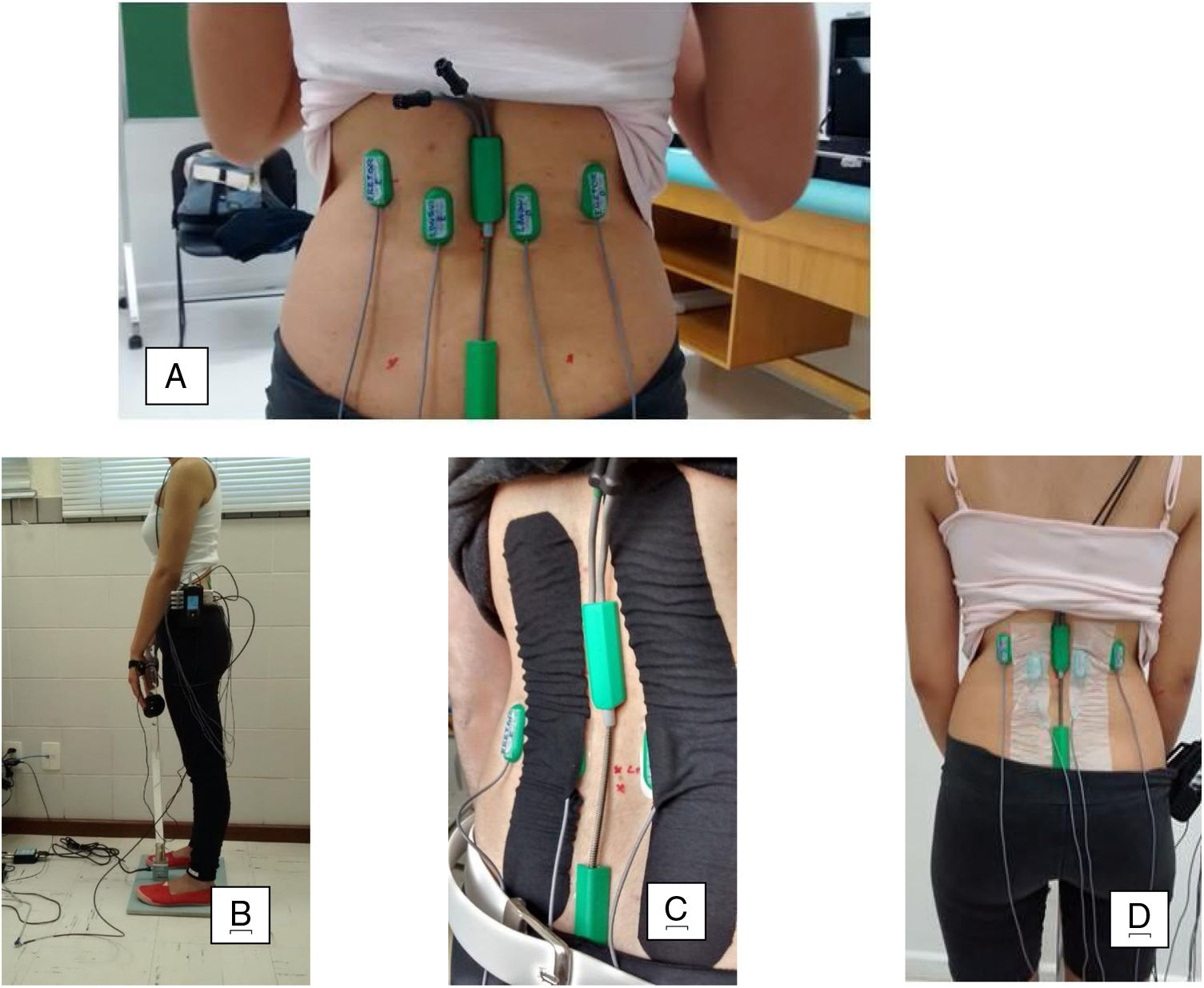
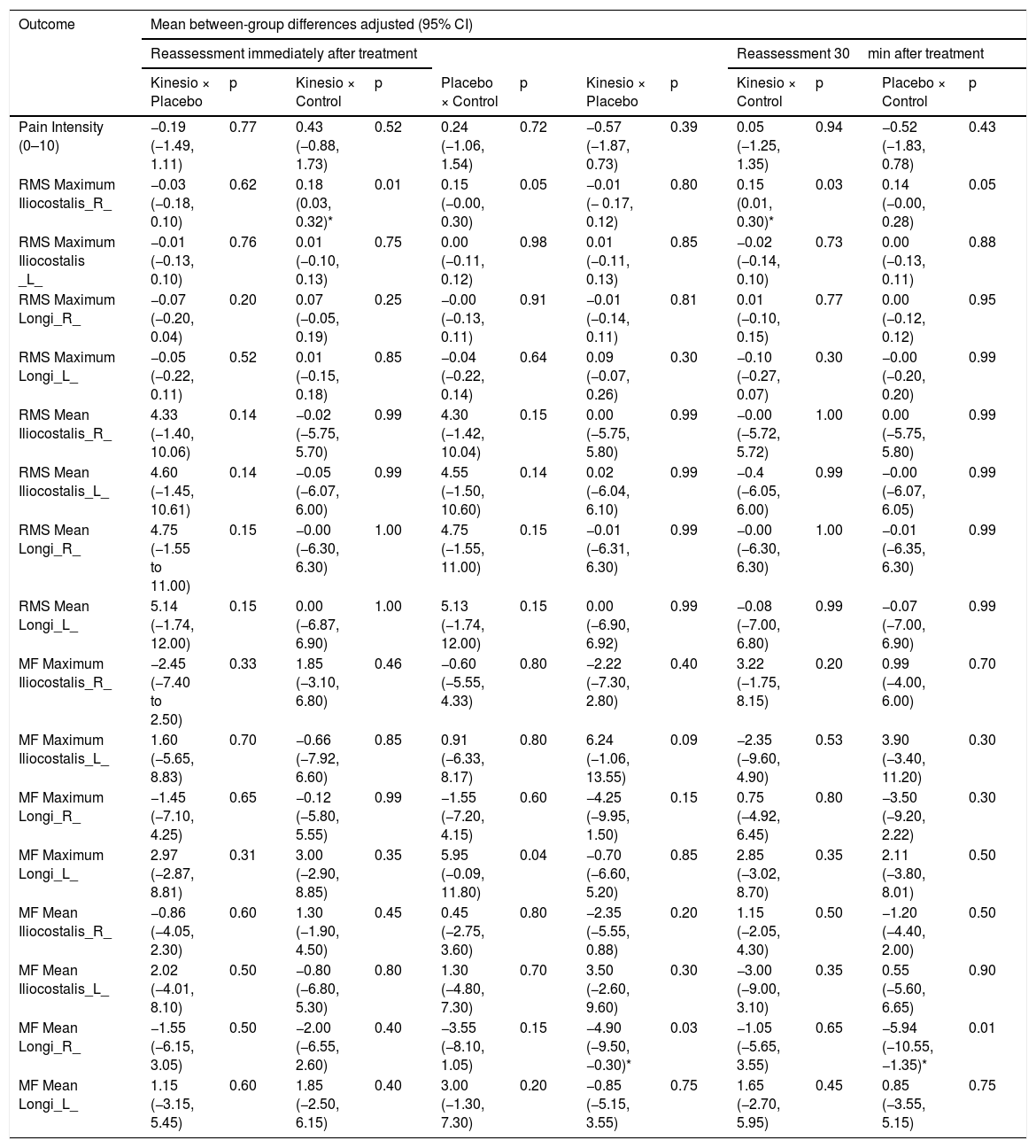
Next, the area to be tested was shaved and cleansed with 70% alcohol. The surface EMG electrodes were applied with T350 double-sided tape, and the electrogoniometer sensors, with T10 double-sided tape. The participants were positioned in slight trunk flexion for the instrumentation: placement of the electrogoniometer and surface EMG electrodes made according to SENIAM26–28 recommendations and placement of the lumbar dynamometry sensors. The EMG electrodes were applied bilaterally on the longissimus, two fingers width from the L1 spinous process and over the iliocostalis at the L2 level (Fig. 1a).29 In addition, an electrogoniometer was positioned between the T12 and S1 vertebrae. One arm of the electrogoniometer was directed caudally and the other cranially with the slightly tensioned spring aligned with the spine (Fig. 1a). The outputs of each channel of the electrogoniometer were previously calibrated according to manufacturer´s instructions to capture the angle variations during data collection.30 Then the participant was positioned in trunk extension, from the position of 30 degrees of trunk flexion measured by a common goniometer (Fig. 1b),31 as well as extension of the cervical spine, elbows, and knees.
Subsequently, the participants received instructions from the therapist to become familiar with the equipment through the trunk extension movement. From that position, the initial evaluation (pre-intervention) was conducted for a maximum voluntary isometric contraction (MVIC) test, in a sustained position for 10s, with the EMG signal synchronized to the electrogoniometer and dynamometer data.32 The first two seconds and the final two seconds were disregarded to minimize signal contamination, therefore only the central six seconds of data collection were used for analysis.
After this pre-intervention assessment, participants were randomized into three treatment groups: Intervention/KT, Placebo, or Control.
EMG processing and data analysisAn electrogoniometer synchronized to the EMG was used to simultaneously capture the range of motion of the thoracolumbar region during the MVIC test on the lumbar dynamometer. The lumbar dynamometer also recorded the mean and maximum trunk muscle strength. The EMG signal was processed and analyzed using the Biometrics EMG software for DataLOG version 8.51. The raw EMG data were captured and the software provided the Root Mean Square (RMS) and Median Frequency (MF) values for the right/left iliocostalis and longissimus muscles. Data were collected for 10s, with the initial and final seconds subsequently trimmed and eliminated. The remaining six-second window was analyzed. The raw EMG signal was used as reference in this study,33 because in situations of the EMG set-up and controlled environment, the use of the non-normalization signal presents good reliability.34
RandomizationThe randomization process was generated by a software program (Excel Office 2010) and performed by a researcher not involved with data collection. Concealed allocation was achieved through the use of sequentially numbered, sealed, and opaque envelopes. After the initial evaluation, the participants were randomly allocated to one of the three groups: Kinesio Taping group; Placebo group, and Control group.
InterventionsKinesio Taping Group: Patients allocated to this group received the Tex Gold Kinesio Taping® bandage according to manufacturer´s instructions.17 The bandage was applied in the shape of an “I” in the direction of insertion (S1 region) to the origin (T12 region), with tension of 10–15% (Paper-off), in the trunk flexion position. This procedure was conducted minutes after instrumentation and pre-intervention assessment. KT was applied over the EMG electrodes monitoring the longissimus muscles at the L1 level (portion of the longissimus dorsi; Fig. 1c).
Placebo Group: Patients allocated to this group had a Missner® surgical tape (5cm in width) placed on the longissimus muscle. The form of application adopted generated folds in the skin, and an “I” shape in the trunk flexion position. The tape was applied over the EMG electrodes, bilaterally to the spine towards the insertion at S1 to the origin at T12, minutes after instrumentation (Fig. 1d).
Control Group: The patients allocated to this group did not receive any type of bandage.
Immediately after randomization and application of the intervention, the participants repeated the maximum voluntary isometric contraction (MVIC) test, in a sustained position for 10s, as performed pre-intervention. The same test was again performed 30min post-intervention with the tape remaining in place.
Outcome measuresThe primary and secondary outcome measures of the study were obtained by an assessor who was unaware of the allocation of patients to their treatment groups by covering their back with a shirt. The primary outcome of the study was the EMG signal of the iliocostalis and longissimus muscles.
The EMG signal was acquired with an eight channel Biometrics DataLog MWX8 wireless data system via SX230–1000 EMG electrodes (Biometrics Ltd., Gwent, UK) with the following specifications: active (single differential), bipolar, reusable surface disk electrodes with eighth order preamplifier and elliptical filter (-60dB at 550Hz), 38mm long, 20mm wide, 0.6mm thick, 10mm diameter discs, 20−450Hz bandpass filter, and third order high pass filter (18dB/octave; 20Hz), CMRR>96dB; noise ratio < 5 μV Input impedance > 1015 Ω Ohms. The connection cables of each channel were previously tagged with the name of the evaluated muscles and the tags remained in place for all data collections. The EMG signal was amplified (K800) and sampled at 1000Hz and data were reported according to the International Society of Electrophysiology and Kinesiology (ISEK) guidelines. A reference electrode (Biometrics R206) was placed on the right wrist.35 In our study, crosstalk was minimized through wireless transmission and the use of surface sensors with amplifiers and internal filters. This choice was based on the small difference in crosstalk when compared to intramuscular electrodes,36 as well as good reliability.37 In addition, the MVIC angle was limited to 30°, minimizing sensor movement. A 20Hz filter was also used, which is above the minimum recommended of 10Hz,38 thus minimizing contamination by up to 47%.39 In the EMG literature, signal normalization is important to eliminate signal variability between evaluations40 and this process depends very much on the type of activity in which signal was collected, e.g., static41 or dynamic condition,42 healthy or unhealthy participants,43 by means of maximum voluntary contraction or not.44 The EMG signal of the right and left iliocostalis and longissimus muscles was captured for signal amplitude analysis (RMS) and Medium Frequency (MF) in the initial evaluation (pre-intervention), after randomization (immediately post-intervention), and 30min post-intervention.
Data collection was performed by three individuals (one therapist and two assessors). The therapist participated in 100% of the EMG data collections. Narayanaswami et al.45 reported the importance of having the same therapist for good reliability with EMG equipment.
The secondary outcome of the study was pain intensity as measured by the 11-point NPRS scored which ranges from 0 (no pain) to 10 (worst possible pain).46 Pain intensity was collected minutes before the initial evaluation (pre-intervention), shortly after randomization (immediately post-intervention), and 30min after randomization (30min post-intervention).
In addition to the collection of the primary and secondary outcomes, RMDQ25,47 was only administered pre-intervention to measure disability at baseline. The questionnaire consists of 24 items that describe activities that patients with LBP may have difficulty performing on a daily basis, where each affirmative answer corresponds to one point on the scale. The final score of the RMDQ was determined by the sum of the values obtained: the higher the score, the greater the disability.25,46 Patients were instructed to complete the statements according to how they felt on the day of the evaluation.
Sample size calculationThe sample calculation of the study was performed to detect a difference of 2.8 units in RMS of the EMG signal with estimated standard deviation of 2.5 units of the EMG signal.48 Assuming a statistical power of 95%, alpha of 5%, and a possible sample loss of up to 20%, a total of 63 participants (21 participants per group) were included.
Statistical analysisThe statistical analysis followed intention-to-treat principles (i.e. patients were analyzed into the groups they were originally allocated to).49 The normality of the data was tested by visual inspection of histograms, and the characterization of the participants was presented using descriptive statistical tests. The primary analysis of interest was the difference in EMG signal between the 3 groups. The between-group differences (effects of treatment) and their respective 95% confidence intervals (CI) were calculated by constructing mixed linear models50 using interaction terms (group versus time). Treatment effects were adjusted for baseline values for all outcomes and for multiple comparisons. Missing data were handled using linear mixed models. The analyses were performed using the software SPSS 19.
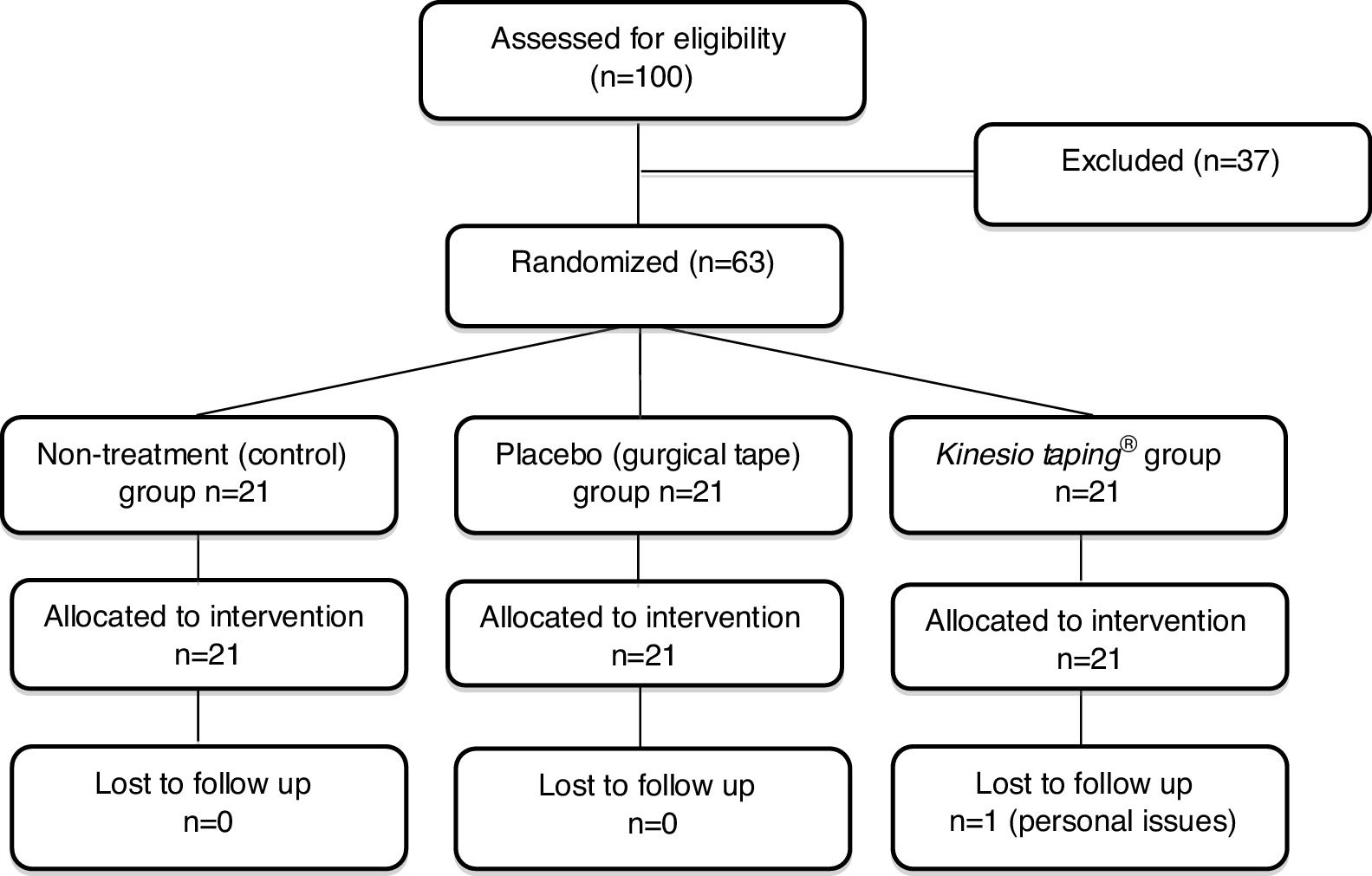
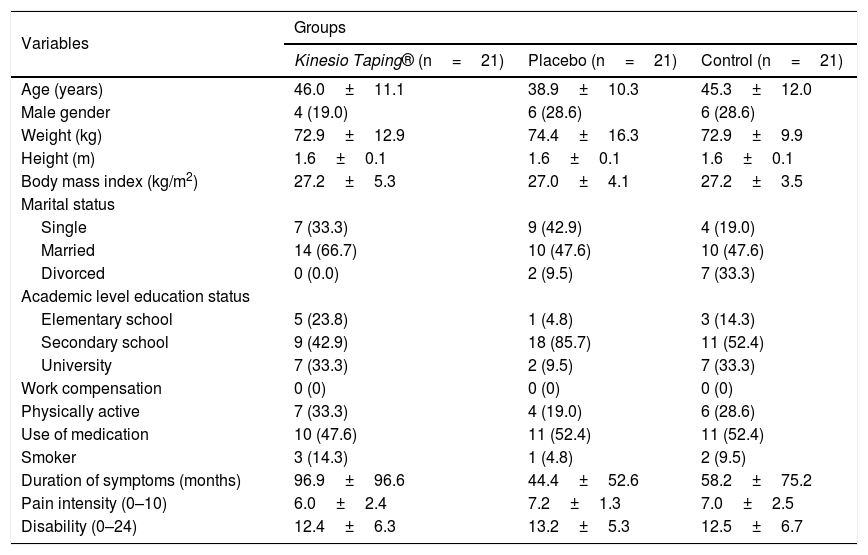
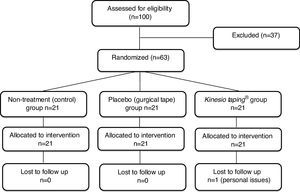
ResultsRecruitment and baseline evaluationFrom a total of 100 potential participants identified between August and December 2016, 63 were considered eligible and were included in the study. Potential participants were excluded due to different ineligibility reasons: age over 60 years (n=25), age under 18 years (n=1), acute non-specific LBP (n=5), previous spinal surgery (n=2), knowledge of the Kinesio Taping® technique (n=2), nerve root involvement (n=1), and knee pain only (n=1). All randomized patients were included in the statistical analysis (Fig. 2). One patient dropped out from the remaining assessments after been assessed at baseline due to personal issues. The characteristics of the included participants in the initial evaluation are described in Table 1. The baseline characteristics of all variables were similar in the three groups.
Characteristics of participants at baseline (n=63).
| Variables | Groups | ||
|---|---|---|---|
| Kinesio Taping® (n=21) | Placebo (n=21) | Control (n=21) | |
| Age (years) | 46.0±11.1 | 38.9±10.3 | 45.3±12.0 |
| Male gender | 4 (19.0) | 6 (28.6) | 6 (28.6) |
| Weight (kg) | 72.9±12.9 | 74.4±16.3 | 72.9±9.9 |
| Height (m) | 1.6±0.1 | 1.6±0.1 | 1.6±0.1 |
| Body mass index (kg/m2) | 27.2±5.3 | 27.0±4.1 | 27.2±3.5 |
| Marital status | |||
| Single | 7 (33.3) | 9 (42.9) | 4 (19.0) |
| Married | 14 (66.7) | 10 (47.6) | 10 (47.6) |
| Divorced | 0 (0.0) | 2 (9.5) | 7 (33.3) |
| Academic level education status | |||
| Elementary school | 5 (23.8) | 1 (4.8) | 3 (14.3) |
| Secondary school | 9 (42.9) | 18 (85.7) | 11 (52.4) |
| University | 7 (33.3) | 2 (9.5) | 7 (33.3) |
| Work compensation | 0 (0) | 0 (0) | 0 (0) |
| Physically active | 7 (33.3) | 4 (19.0) | 6 (28.6) |
| Use of medication | 10 (47.6) | 11 (52.4) | 11 (52.4) |
| Smoker | 3 (14.3) | 1 (4.8) | 2 (9.5) |
| Duration of symptoms (months) | 96.9±96.6 | 44.4±52.6 | 58.2±75.2 |
| Pain intensity (0–10) | 6.0±2.4 | 7.2±1.3 | 7.0±2.5 |
| Disability (0–24) | 12.4±6.3 | 13.2±5.3 | 12.5±6.7 |
Categorical variables are expressed in n (%) and continuous variables as mean±standard deviation.
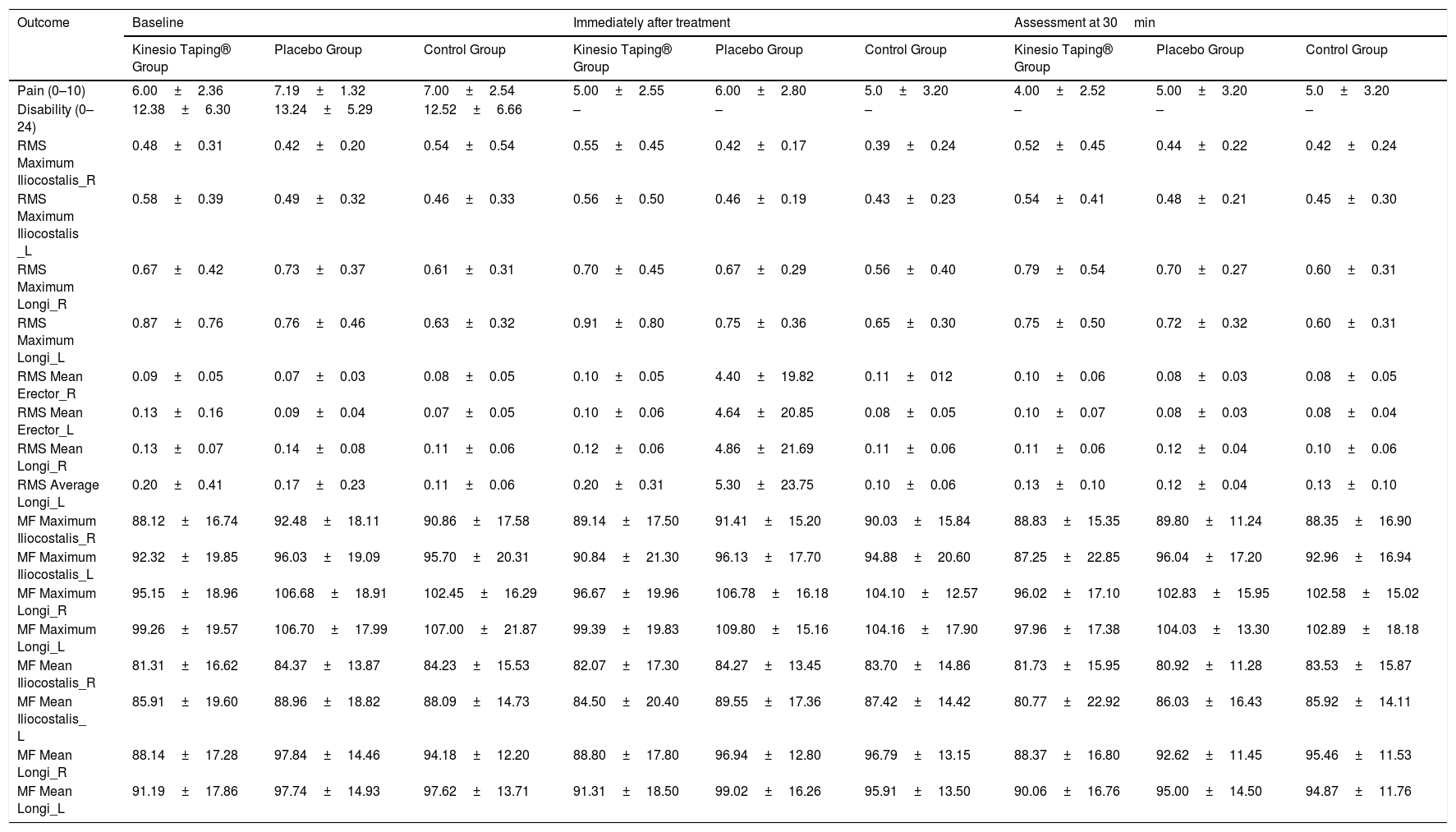
Table 2 presents the descriptive data of the primary and secondary variables at all time points. There was no significant change in muscle activity of the iliocostalis and longissimus muscles as measured by EMG amplitude (RMS) and frequency (MF) with the use of Kinesio Taping. Of the 96 statistical comparisons (Table 3), a statistically significant difference in EMG signal was only observed for the RMS max of the right iliocostalis muscle and the medium frequency (MF) of the right longissimus muscle in favor of the Kinesio Taping group compared to the placebo (surgical tape) and control group.
Outcomes at baseline, after Intervention, and after 30min.
| Outcome | Baseline | Immediately after treatment | Assessment at 30min | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Kinesio Taping® Group | Placebo Group | Control Group | Kinesio Taping® Group | Placebo Group | Control Group | Kinesio Taping® Group | Placebo Group | Control Group | |
| Pain (0–10) | 6.00±2.36 | 7.19±1.32 | 7.00±2.54 | 5.00±2.55 | 6.00±2.80 | 5.0±3.20 | 4.00±2.52 | 5.00±3.20 | 5.0±3.20 |
| Disability (0–24) | 12.38±6.30 | 13.24±5.29 | 12.52±6.66 | – | – | – | – | – | – |
| RMS Maximum Iliocostalis_R | 0.48±0.31 | 0.42±0.20 | 0.54±0.54 | 0.55±0.45 | 0.42±0.17 | 0.39±0.24 | 0.52±0.45 | 0.44±0.22 | 0.42±0.24 |
| RMS Maximum Iliocostalis _L | 0.58±0.39 | 0.49±0.32 | 0.46±0.33 | 0.56±0.50 | 0.46±0.19 | 0.43±0.23 | 0.54±0.41 | 0.48±0.21 | 0.45±0.30 |
| RMS Maximum Longi_R | 0.67±0.42 | 0.73±0.37 | 0.61±0.31 | 0.70±0.45 | 0.67±0.29 | 0.56±0.40 | 0.79±0.54 | 0.70±0.27 | 0.60±0.31 |
| RMS Maximum Longi_L | 0.87±0.76 | 0.76±0.46 | 0.63±0.32 | 0.91±0.80 | 0.75±0.36 | 0.65±0.30 | 0.75±0.50 | 0.72±0.32 | 0.60±0.31 |
| RMS Mean Erector_R | 0.09±0.05 | 0.07±0.03 | 0.08±0.05 | 0.10±0.05 | 4.40±19.82 | 0.11±012 | 0.10±0.06 | 0.08±0.03 | 0.08±0.05 |
| RMS Mean Erector_L | 0.13±0.16 | 0.09±0.04 | 0.07±0.05 | 0.10±0.06 | 4.64±20.85 | 0.08±0.05 | 0.10±0.07 | 0.08±0.03 | 0.08±0.04 |
| RMS Mean Longi_R | 0.13±0.07 | 0.14±0.08 | 0.11±0.06 | 0.12±0.06 | 4.86±21.69 | 0.11±0.06 | 0.11±0.06 | 0.12±0.04 | 0.10±0.06 |
| RMS Average Longi_L | 0.20±0.41 | 0.17±0.23 | 0.11±0.06 | 0.20±0.31 | 5.30±23.75 | 0.10±0.06 | 0.13±0.10 | 0.12±0.04 | 0.13±0.10 |
| MF Maximum Iliocostalis_R | 88.12±16.74 | 92.48±18.11 | 90.86±17.58 | 89.14±17.50 | 91.41±15.20 | 90.03±15.84 | 88.83±15.35 | 89.80±11.24 | 88.35±16.90 |
| MF Maximum Iliocostalis_L | 92.32±19.85 | 96.03±19.09 | 95.70±20.31 | 90.84±21.30 | 96.13±17.70 | 94.88±20.60 | 87.25±22.85 | 96.04±17.20 | 92.96±16.94 |
| MF Maximum Longi_R | 95.15±18.96 | 106.68±18.91 | 102.45±16.29 | 96.67±19.96 | 106.78±16.18 | 104.10±12.57 | 96.02±17.10 | 102.83±15.95 | 102.58±15.02 |
| MF Maximum Longi_L | 99.26±19.57 | 106.70±17.99 | 107.00±21.87 | 99.39±19.83 | 109.80±15.16 | 104.16±17.90 | 97.96±17.38 | 104.03±13.30 | 102.89±18.18 |
| MF Mean Iliocostalis_R | 81.31±16.62 | 84.37±13.87 | 84.23±15.53 | 82.07±17.30 | 84.27±13.45 | 83.70±14.86 | 81.73±15.95 | 80.92±11.28 | 83.53±15.87 |
| MF Mean Iliocostalis_ L | 85.91±19.60 | 88.96±18.82 | 88.09±14.73 | 84.50±20.40 | 89.55±17.36 | 87.42±14.42 | 80.77±22.92 | 86.03±16.43 | 85.92±14.11 |
| MF Mean Longi_R | 88.14±17.28 | 97.84±14.46 | 94.18±12.20 | 88.80±17.80 | 96.94±12.80 | 96.79±13.15 | 88.37±16.80 | 92.62±11.45 | 95.46±11.53 |
| MF Mean Longi_L | 91.19±17.86 | 97.74±14.93 | 97.62±13.71 | 91.31±18.50 | 99.02±16.26 | 95.91±13.50 | 90.06±16.76 | 95.00±14.50 | 94.87±11.76 |
RMS=Root Mean Square; MF=Medium Frequency ; R=Right Side ; L=Left Side; Longi=Longissimus. Data are mean±standard deviation.
Effects of interventions.
| Outcome | Mean between-group differences adjusted (95% CI) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reassessment immediately after treatment | Reassessment 30min after treatment | |||||||||||
| Kinesio × Placebo | p | Kinesio × Control | p | Placebo × Control | p | Kinesio × Placebo | p | Kinesio × Control | p | Placebo × Control | p | |
| Pain Intensity (0–10) | −0.19 (−1.49, 1.11) | 0.77 | 0.43 (−0.88, 1.73) | 0.52 | 0.24 (−1.06, 1.54) | 0.72 | −0.57 (−1.87, 0.73) | 0.39 | 0.05 (−1.25, 1.35) | 0.94 | −0.52 (−1.83, 0.78) | 0.43 |
| RMS Maximum Iliocostalis_R_ | −0.03 (−0.18, 0.10) | 0.62 | 0.18 (0.03, 0.32)* | 0.01 | 0.15 (−0.00, 0.30) | 0.05 | −0.01 (− 0.17, 0.12) | 0.80 | 0.15 (0.01, 0.30)* | 0.03 | 0.14 (−0.00, 0.28) | 0.05 |
| RMS Maximum Iliocostalis _L_ | −0.01 (−0.13, 0.10) | 0.76 | 0.01 (−0.10, 0.13) | 0.75 | 0.00 (−0.11, 0.12) | 0.98 | 0.01 (−0.11, 0.13) | 0.85 | −0.02 (−0.14, 0.10) | 0.73 | 0.00 (−0.13, 0.11) | 0.88 |
| RMS Maximum Longi_R_ | −0.07 (−0.20, 0.04) | 0.20 | 0.07 (−0.05, 0.19) | 0.25 | −0.00 (−0.13, 0.11) | 0.91 | −0.01 (−0.14, 0.11) | 0.81 | 0.01 (−0.10, 0.15) | 0.77 | 0.00 (−0.12, 0.12) | 0.95 |
| RMS Maximum Longi_L_ | −0.05 (−0.22, 0.11) | 0.52 | 0.01 (−0.15, 0.18) | 0.85 | −0.04 (−0.22, 0.14) | 0.64 | 0.09 (−0.07, 0.26) | 0.30 | −0.10 (−0.27, 0.07) | 0.30 | −0.00 (−0.20, 0.20) | 0.99 |
| RMS Mean Iliocostalis_R_ | 4.33 (−1.40, 10.06) | 0.14 | −0.02 (−5.75, 5.70) | 0.99 | 4.30 (−1.42, 10.04) | 0.15 | 0.00 (−5.75, 5.80) | 0.99 | −0.00 (−5.72, 5.72) | 1.00 | 0.00 (−5.75, 5.80) | 0.99 |
| RMS Mean Iliocostalis_L_ | 4.60 (−1.45, 10.61) | 0.14 | −0.05 (−6.07, 6.00) | 0.99 | 4.55 (−1.50, 10.60) | 0.14 | 0.02 (−6.04, 6.10) | 0.99 | −0.4 (−6.05, 6.00) | 0.99 | −0.00 (−6.07, 6.05) | 0.99 |
| RMS Mean Longi_R_ | 4.75 (−1.55 to 11.00) | 0.15 | −0.00 (−6.30, 6.30) | 1.00 | 4.75 (−1.55, 11.00) | 0.15 | −0.01 (−6.31, 6.30) | 0.99 | −0.00 (−6.30, 6.30) | 1.00 | −0.01 (−6.35, 6.30) | 0.99 |
| RMS Mean Longi_L_ | 5.14 (−1.74, 12.00) | 0.15 | 0.00 (−6.87, 6.90) | 1.00 | 5.13 (−1.74, 12.00) | 0.15 | 0.00 (−6.90, 6.92) | 0.99 | −0.08 (−7.00, 6.80) | 0.99 | −0.07 (−7.00, 6.90) | 0.99 |
| MF Maximum Iliocostalis_R_ | −2.45 (−7.40 to 2.50) | 0.33 | 1.85 (−3.10, 6.80) | 0.46 | −0.60 (−5.55, 4.33) | 0.80 | −2.22 (−7.30, 2.80) | 0.40 | 3.22 (−1.75, 8.15) | 0.20 | 0.99 (−4.00, 6.00) | 0.70 |
| MF Maximum Iliocostalis_L_ | 1.60 (−5.65, 8.83) | 0.70 | −0.66 (−7.92, 6.60) | 0.85 | 0.91 (−6.33, 8.17) | 0.80 | 6.24 (−1.06, 13.55) | 0.09 | −2.35 (−9.60, 4.90) | 0.53 | 3.90 (−3.40, 11.20) | 0.30 |
| MF Maximum Longi_R_ | −1.45 (−7.10, 4.25) | 0.65 | −0.12 (−5.80, 5.55) | 0.99 | −1.55 (−7.20, 4.15) | 0.60 | −4.25 (−9.95, 1.50) | 0.15 | 0.75 (−4.92, 6.45) | 0.80 | −3.50 (−9.20, 2.22) | 0.30 |
| MF Maximum Longi_L_ | 2.97 (−2.87, 8.81) | 0.31 | 3.00 (−2.90, 8.85) | 0.35 | 5.95 (−0.09, 11.80) | 0.04 | −0.70 (−6.60, 5.20) | 0.85 | 2.85 (−3.02, 8.70) | 0.35 | 2.11 (−3.80, 8.01) | 0.50 |
| MF Mean Iliocostalis_R_ | −0.86 (−4.05, 2.30) | 0.60 | 1.30 (−1.90, 4.50) | 0.45 | 0.45 (−2.75, 3.60) | 0.80 | −2.35 (−5.55, 0.88) | 0.20 | 1.15 (−2.05, 4.30) | 0.50 | −1.20 (−4.40, 2.00) | 0.50 |
| MF Mean Iliocostalis_L_ | 2.02 (−4.01, 8.10) | 0.50 | −0.80 (−6.80, 5.30) | 0.80 | 1.30 (−4.80, 7.30) | 0.70 | 3.50 (−2.60, 9.60) | 0.30 | −3.00 (−9.00, 3.10) | 0.35 | 0.55 (−5.60, 6.65) | 0.90 |
| MF Mean Longi_R_ | −1.55 (−6.15, 3.05) | 0.50 | −2.00 (−6.55, 2.60) | 0.40 | −3.55 (−8.10, 1.05) | 0.15 | −4.90 (−9.50, −0.30)* | 0.03 | −1.05 (−5.65, 3.55) | 0.65 | −5.94 (−10.55, −1.35)* | 0.01 |
| MF Mean Longi_L_ | 1.15 (−3.15, 5.45) | 0.60 | 1.85 (−2.50, 6.15) | 0.40 | 3.00 (−1.30, 7.30) | 0.20 | −0.85 (−5.15, 3.55) | 0.75 | 1.65 (−2.70, 5.95) | 0.45 | 0.85 (−3.55, 5.15) | 0.75 |
RMS=Root Mean Square; MF=Medium Frequency; R=Right Side; L=Left side; Longi=Longissimus; *Significant difference (p<0.05).
We aimed to determine whether KT would change the EMG signal intensity of the iliocostalis and longissimus muscles of patients with chronic non-specific LBP. Compared to control or placebo, KT reduced EMG signal in only 4 of 96 statistical comparisons, which suggests that these findings are spurious due to the high number of statistical comparisons. No between-group differences were identified for the immediate reduction in pain intensity.
To our knowledge, this is the first trial that aimed to identify the effects of Kinesio Taping®, compared to a placebo treatment, on the EMG signal intensity of lumbar muscles of patients with LBP. The study’s strengths were 1) control of the main sources of bias in clinical trials, such as concealed allocation, assessor blinding, and intention-to-treat analysis,51 2) rigorous use of all SENIAM guidelines,52 and 3) strict adherence to ISEK (International Society of Electrophysiology and Kinesiology)38 guidelines.
However, our study has some limitations. First of all, we did not include only patients with high levels of muscle activity as we assumed that this is very common in patients with LBP. Due the nature of the interventions, it was not possible to blind the therapist and patients. Another limitation was that signal contamination could not be completely eliminated. Also, the tapes were placed on top of the EMG electrodes, however these electrodes were very small and it is unlikely to influence the outcomes. Although randomization generated similar groups in most variables, the duration of symptoms among the three treatment arms were different. Nevertheless all participants from this trial were patients with long-lasting chronic LBP. There is no evidence that the prognosis of a patient with 97 months (e.g. a patient from the KT group) would be different compared to a patient from the placebo group with 44 months of symptoms. Clinically speaking, it is unlikely that a physical therapist would considered these patients as separate subgroups. The most important systematic review53 on the prognosis of patients with chronic LBP only provides data up to 12 months. Therefore, we do not have data to support or refute the hypothesis that these differences in duration of symptoms could influence the outcomes of the trial. Finally, we conducted quite a large number of statistical comparisons. Therefore the chances of type 1 errors (i.e. false positive findings) were very likely. We used linear mixed models to test between group differences adjusted for baseline estimates even knowing that these differences at baseline were not clinically relevant. Although we did not use corrections such as Bonferroni corrections, we ended up getting most regression coefficients not statistically significant. Only four of 96 estimates (4.2%) were statistically significant, very likely reflecting spurious findings.
Among all clinical trials on the efficacy of Kinesio Taping® performed in patients with chronic LBP14,54–56 three used EMG as an outcome.15,22,23,57 In these trials, no clinically important results was found for Kinesio Taping®, which is consistent with our findings. Regarding the primary outcome measure (EMG signal), the difference between the treatment, control, and placebo groups was not statistically significant for most of the comparisons. Fong et al.23 also did not observe a significant change in the EMG signal intensity of the lumbar multifidus muscles after the use of Kinesio Taping® compared to the control group that received no intervention. Similar results were observed from Paoloni et al.15. who measured EMG signals on a three-arm trial comparing exercises versus KT versus KT plus exercises in patients with LBP. These authors15 also did not find correlations among changes in EMG signals and changes in pain intensity. Conversely, a very small (n=20) and low quality trial conducted by Bae et al.22 concluded that KT changed EMG findings compared to “ordinary physical therapy”. From a purely statistical point of view, the few statistically significant findings may have been random and caused by type I statistical errors.58,59 Therefore, the prescription of KT with the goal to reduce muscle activation does not seem justified.
There is a large amount of clinical evidence, including two meta-analyses,13,60 four systematic reviews56,61–63 in musculoskeletal conditions, and one for LBP64 that consistently conclude that the effects of Kinesio Taping® for these patients are not better than placebo.65 Therefore, the current evidence does not support the use of KT in patients with chronic non-specific LBP. On the other hand, the number of studies on the mechanisms of action of KT in patients with musculoskeletal conditions is still very small. Therefore, more research is needed in this area.
We conclude that KT did not change the EMG signal intensity of the longissimus and iliocostalis muscles or reduced pain intensity in patients with chronic LBP. This specific proposed mechanism of action of this intervention was not supported by the findings of this trial.
AcknowledgementThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The first author of this manuscript received a scholarship from CAPES – Brazil (finance code 001).
Conflict of interestThe authors declare that there is no conflict of interest.