Traumatic Brain Injury (TBI) survivors often experience long-term impairments that might decrease their quality of life and functional independence.
ObjectiveThis study aimed to identify predictors of functional recovery after severe TBI in Brazil.
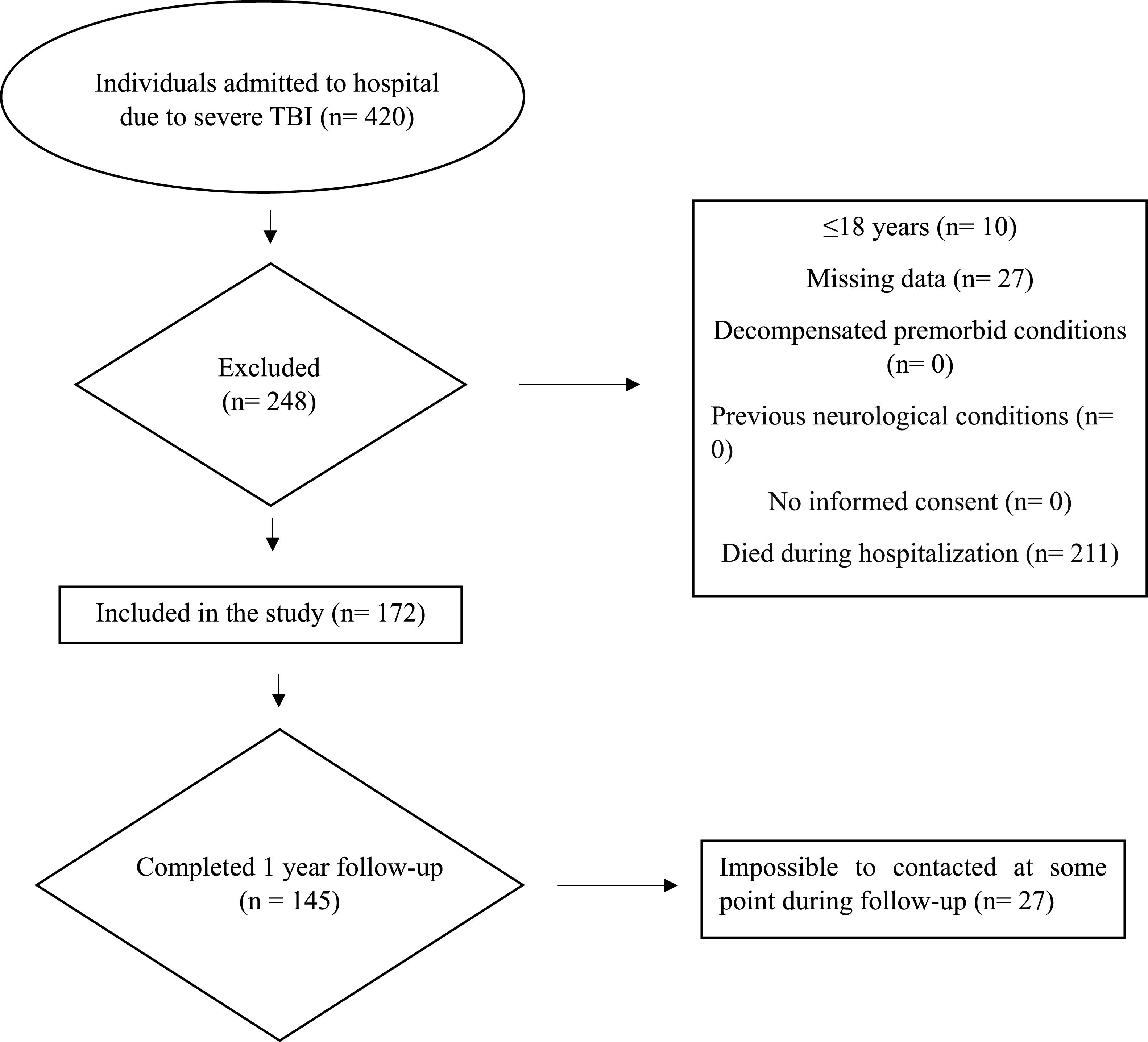
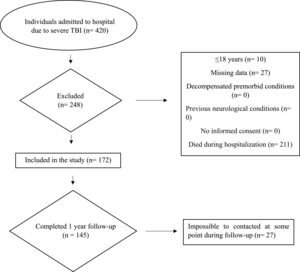
MethodsA prospective observational cohort study was conducted at a trauma referral hospital between May 2021 and May 2022. Individuals with severe TBI (sTBI), defined as having a Glasgow Coma Scale (GCS) score of ≤8 at admission or within 72 h due to head trauma-related causes, were included. Functional recovery was assessed using the Glasgow Outcome Scale-Extended (GOSE) at discharge and 3, 6, and 12 months post-injury. Data on age, sex, cause of injury, GCS scores at admission, Injury Severity Score (ISS), pupillary alterations, decompressive craniectomy, days of mechanical ventilation support (MVS), and education level were extracted from electronic records. Of 172 patients, 145 completed follow-ups.
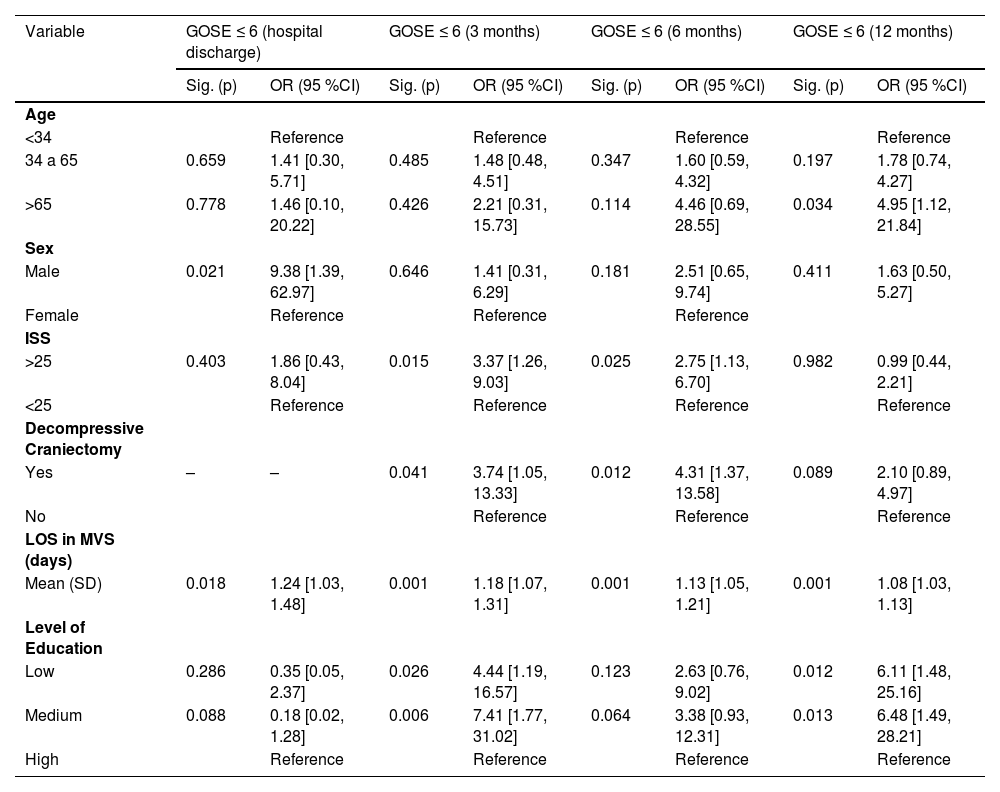
ResultsPatients were mostly male (84.5 %), with low education levels (55.2 %) and an average age of 45. Male sex (OR=9.38, 95 %CI: 1.39, 62.97) and more days of MVS (OR=1.41, 95 %CI: 1.03, 1.48) predicted poor outcomes at discharge. At 3 months, ISS >25 (OR=3.37, 95 %CI: 1.26, 9.03), decompressive craniectomy (OR=3.74, 95 %CI: 1.05, 13.33), more days of MVS (OR=1.18, 95 %CI: 1.07, 1.31), and low (OR=4.44, 95 %CI: 1.19, 16.57) or medium (OR=7.41, 95 %CI: 1.77, 31.02) education levels predicted poorer functional outcomes. At 6 months, decompressive craniectomy (OR=4.31, 95 %CI: 1.37, 13.58), more days of MVS (OR=1.13, 95 %CI: 1.05, 1.21), and a GOSE score ≤6 at discharge were associated with unfavorable functional outcome. At 12 months, age >65 (OR=4.95, 95 %CI: 1.12, 21.84), more days of MVS (OR=1.08, 95 %CI: 1.03, 1.13), and low (OR=6.11, 95 %CI: 1.48, 25.16) or medium (OR=6.48, 95 %CI: 1.49, 28.21) education levels predicted poorer functional recovery.
ConclusionFunctional recovery after severe TBI in a low- to middle-income setting is influenced by clinical and sociodemographic factors.
Traumatic brain injury (TBI) is the leading cause of disability, limitations in daily living activities, and social participation among the young population worldwide.1–2 After TBI, low- to middle-income countries (LMICs) are known to have higher rates of hospital mortality and long-term functional impairments compared to high-income countries (HICs).3–5 Globally, studies confirm that individuals with milder TBI, higher education levels, and shorter stays in the Intensive Care Unit (ICU) have significantly higher survival rates and more favorable functional outcomes.6 Conversely, advanced age, lower Glasgow Coma Scale (GCS) scores, and the presence of subdural hematoma and/or intraventricular hemorrhage on cranial computed tomography (CT) scans are strongly associated with higher hospital mortality rates and long-term disabilities.7–9
In Brazil, TBI represents a significant and increasing public health concern, reflecting the challenges faced by LMICs.10–11 Its rising incidence is mainly related to socioeconomic factors and violence.12–13 Recent data report >131,000 TBI-related hospitalizations annually, with an incidence of 65.5 per 100,000 inhabitants14 and an estimated annual cost of $70 million.15–16 Functional outcomes are often poor as 25–50 % of survivors have unfavorable recovery six months post-injury,17 20 % remain dependent one year after severe TBI,18 and 68 % continue to experience disabilities five years later.19 Despite this burden, research on predictors of functional outcomes in the Brazilian population remains limited.
There are few studies in Brazil that investigated multiple predictors of functional outcomesat 617 and 12 months20,21,18,22 after sTBI. The findings suggested that low GCS scores,17 advanced age, trauma severity, and length of hospitalization were independent variables associated with functional outcomes.17,20 However, these studies present significant limitations such as a mixed sample composed of TBI and stroke cases,17 sample composed of only male individuals, and without differentiation of injury severity levels.20
Due to this gap in literature the functional trajectory after hospital discharge and many of the factors that influence the outcomes of severe TBI are still unknown. Elucidating the associations between clinical and sociodemographic variables and long-term functional outcomes in this population will be useful in guiding public policies for prevention and health promotion at national and regional levels. Based on this rationale, the hypothesis of this study is that clinical and sociodemographic factors are significant predictors of functional outcomes in individuals with sTBI within the first-year post-injury. Therefore, the present study aimed to investigate multiple predictors of functional outcomes within the first year after sTBI in Brazil.
MethodsThis study was approved by the Research Ethics Committee of the Universidade Federal do Espírito Santo under the protocol number of 4.222.002. The Strengthening the reporting of observational studies in epidemiology (STROBE) guidelines were used to ensure the reporting of the study.
Study designThis is a prospective cohort conducted at a trauma referral hospital in Brazil. Further details of the procedures are described in our previously published protocol paper.23
All patients with severe TBI admitted to the Hospital Estadual de Urgência e Emergência (HEUE), between May 2021 and May 2022, were invited to participate. The main outcome of interest was functional recovery in the first year after sTBI.
Participants were recruited by a trained researcher within 72 h of admission. Written consent was obtained from the patient or the closest family member present. Once participants agreed to join the study, the independent variables were obtained from electronic records. The functional outcomes of the patients were assessed using the Glasgow Outcome Scale-Extended (GOSE) in person at hospital discharge and by phone at 3, 6, and 12 months later.
ParticipantsAll patients aged ≥18 years admitted to the emergency room with a clinical diagnosis of severe sTBI within 72 h who survived hospitalization were eligible. TBI was considered severe if the patient had a Glasgow Coma Scale score of ≤8 at the time of admission or within 72 h due to head trauma-related causes.24 Exclusion criteria consisted of individuals under 18 years old; death during hospitalization, patients with missing age, sex, admission GCS score or time of admission in their medical records; decompensated premorbid conditions as pre-existing heart failure, chronic kidney diseases, and cancers; previous neurological conditions such as prior stroke, TBI, or spinal cord injury (SCI); and individuals who did not sign the informed consent form, either themselves or through their legal guardian.
MeasuresOutcome of interestFunctional recovery was assessed using GOSE in person at hospital discharge and by telephone at 3, 6, and 12 months after sTBI. Assessments were performed by a single trained investigator. The GOSE is a well validated and the most common scale used to assess functional outcomes of patients with TBI.25 It is applied through a structured interview, which can be conducted in person or by phone, with the patient or a close family member if the patient cannot comprehend or communicate sufficiently well.26 GOSE scores range from 1 to 8: full recovery (8 points); good recovery (7 points); upper moderate disability (6 points); lower moderate disability (5 points); upper severe disability (4 points); lower severe disability (3 points); persistent vegetative state (2 points); and death (1 point).27 The GOSE has high test-retest reliability (KW = 0.92) and good inter-rater reliability (KW = 0.84).28–29 For analysis purposes, the patients were classified into two groups: unfavorable functional recovery (GOSE of 6 or less) and good recovery (GOSE of 7 or 8).
PredictorsAge, sex, cause of injury, GCS scores at hospital admission, pupillary alterations, performance of decompressive craniectomy, and length of stay on mechanical ventilation were obtained from electronic records. Pupils were considered abnormal if described in the medical record as mydriatic, miotic, or anisocoric at the time of hospital admission. The severity of injury was determined by calculating the Injury Severity Score (ISS), based on the Abbreviated Injury Scale (AIS).30 Injury severity was classified as severe when ISS was >25 and as mild or moderate when ISS was ≤25. ISS has been extensively correlated with mortality, morbidity, and hospital stay after TBI.30 It has high interrater reliability, with a kappa of 0.71 (95 %CI: 0.60, 0.82).31 Level of education was obtained through a personal interview with the patient or their guardian at the time of assessment during hospital discharge. It was analyzed as a categorical variable with three distinct categories: low, medium, and high. The low level of education included individuals who had not studied or had not completed primary education. The medium level encompassed those who had completed primary education but had not finished secondary education. The high level comprised individuals who had completed secondary education and those who had pursued higher education.
Statistical analysisDescriptive statistics were performed using means and standard deviation (SD) for continuous variables and proportions for categorical variables. For the functional outcome, binomial logistic regression analysis was performed for each variable individually to identify possible predictors. Variables considered statistically significant were analyzed in a final binomial logistic regression model, with significance at p ≤ 0.05. The magnitude of the association between functional outcome and the predictor variables was measured by the odds ratio (OR) and the respective 95 % confidence interval was reported for each predictor. Analyzes were performed using IBM SPSS Statistics for Windows version 21 (IBM Corp., Chicago, IL).
ResultsBetween May 2021 and May 2022, 420 patients with sTBI were admitted to the State Hospital of Urgency and Emergency (HEUE). After applying the enrollment criteria, 172 (45 %) patients were included in the study. During follow-up, 27 individuals could not be contacted. Consequently, 145 individuals completed the 1-year follow-up (Fig. 1). Mortality rate due to sTBI within the first year was 15 % (n = 22) and 123 patients survived the first year after sTBI.
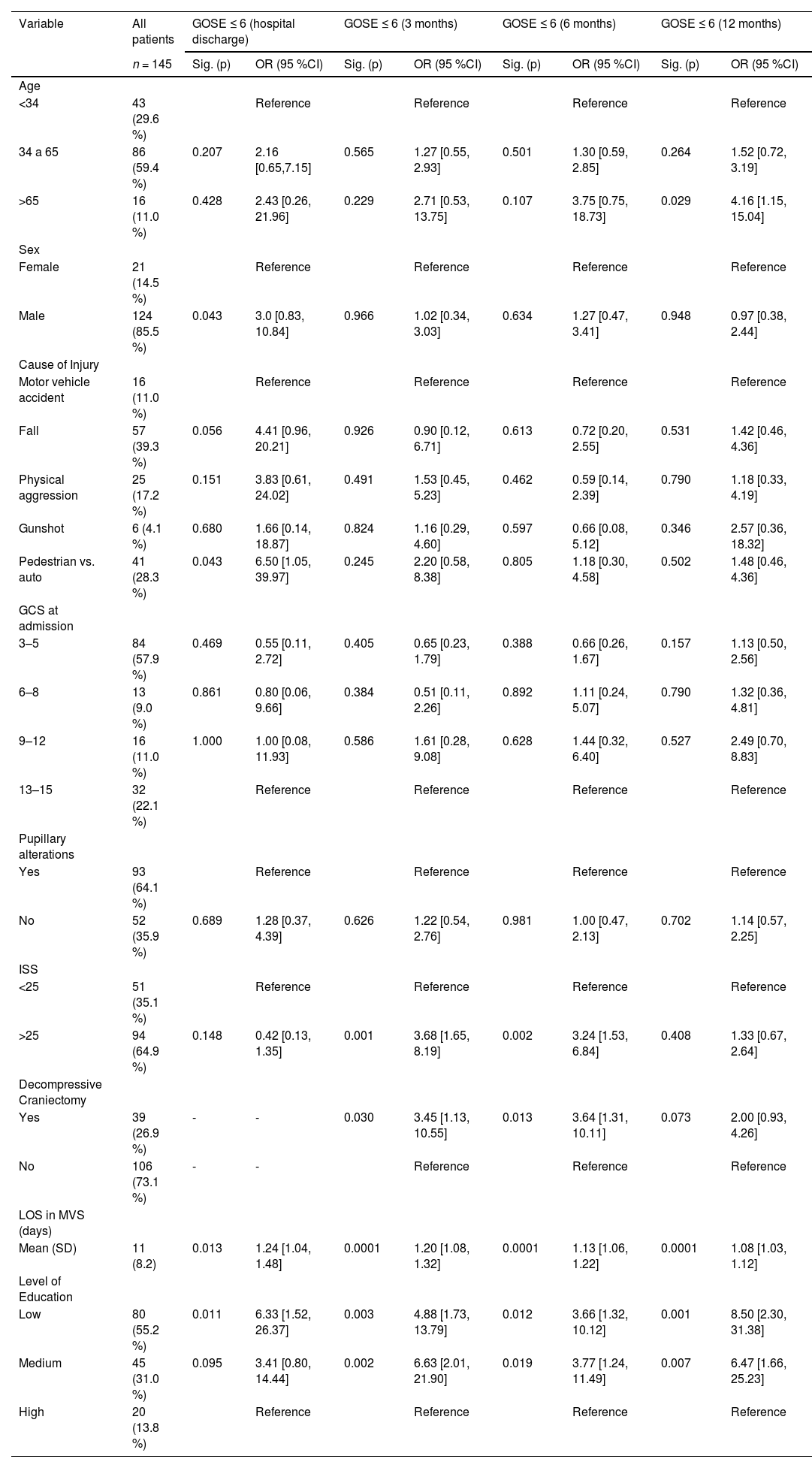
The 145 patients included in the functional outcome analysis were mostly male (85 %) with an average age of 45 (SD: 11) years. The principal causes of injury were falls (39.3 %) and being struck by a vehicle (pedestrian vs. auto) with 28.3 %. The majority of patients (57.9 %) had GCS scores ranging between 3 and 5 at hospital admission. Of the 145 individuals, 80 (55 %) had a low level of education and they spent an average of 11 (SD: ±8) days under mechanical ventilation assistance. Other sociodemographic and clinical characteristics are described in Table 1.
Sociodemographic and clinical characteristics of individuals who completed 12 months follow-up and binary logistic regression for each predictor.
GCS, Glasgow Coma Scale; GOSE, Glasgow Outcome Scale-Extended; ISS, Injury Severity Score; LOS, length of stay;.
MVS, mechanical ventilation support.
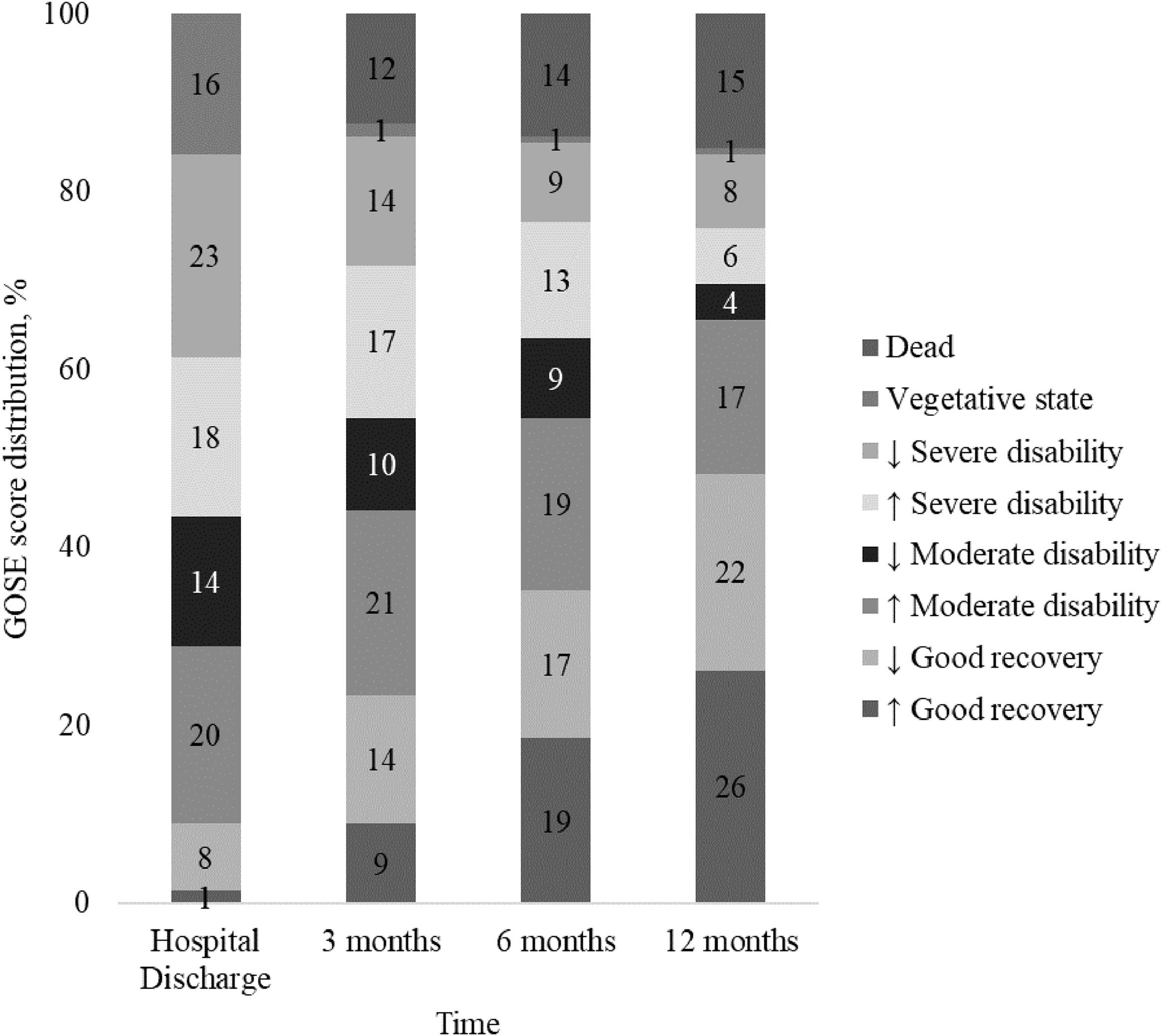
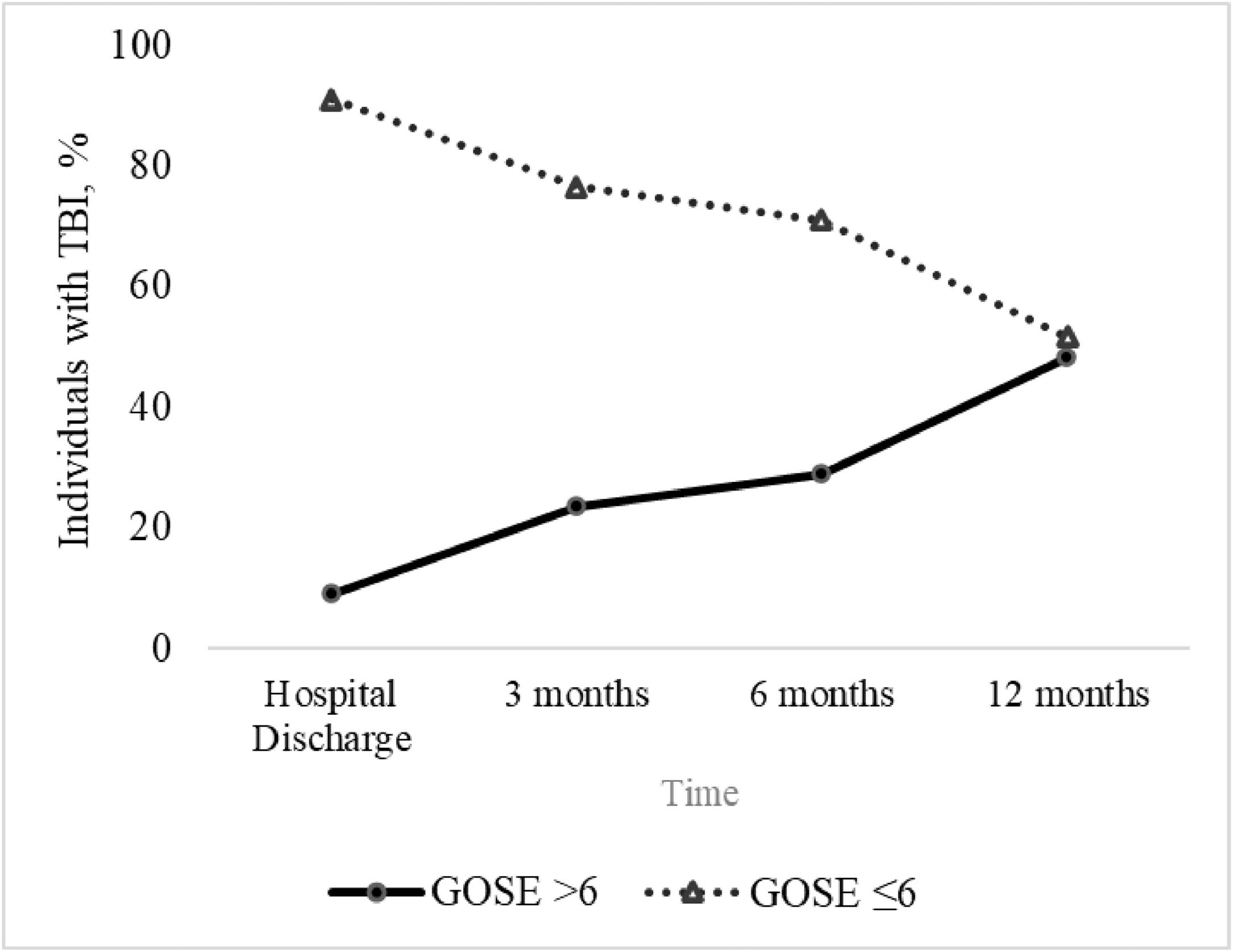
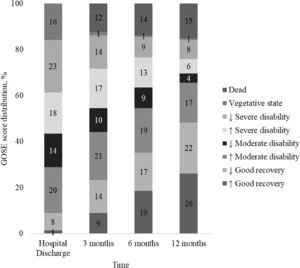
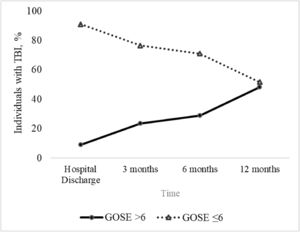
At hospital discharge, the majority (91 %) of patients had an unfavorable functional outcome (GOSE ≤ 6) and of these, 23 % were classified as lower severe disability while only (9 %) had a good functional recovery (GOSE of 7 or 8). Over the first year, this proportion reversed as the number of patients with GOSE scores ≤ 6 decreased and those with scores > 6 increased. Three months after TBI, the favorable outcome increased to 24 % of the sample, while unfavorable outcome decreased to 76 %. At 6 months, 38 % of patients had a favorable functional outcome, while 62 % had an unfavorable outcome. One year after the trauma, 48 % of patients had an unfavorable functional outcome, and 52 % had a good functional recovery. GOSE scores at hospital discharge, and at 3, 6, and 12 months after severe TBI and the trajectory of functional recovery over 12 months after severe TBI, are detailed in Figs. 2 and 3.
After individual analysis of the variables, age, sex, ISS score, performance of decompressive craniectomy, length of stay on mechanical ventilation support (MVS), and level of education were selected as potential predictors of mortality (Table 1).
In the final logistic regression model, being male (OR = 9.38, 95 %: CI 1.39, 62.97) and a greater length of stay on MVS (OR = 1.24, 95 % CI: 1.03, 1.48) were variables associated with a higher risk of an unfavorable functional outcome at hospital discharge. At 3 months post-trauma, an ISS score > 25 (OR = 3.37, 95 % CI: 1.26, 9.03), the performance of decompressive craniectomy (OR = 3.74, 95 % CI: 1.05, 13.33), greater length of stay on MVS (OR = 1.19 95 % CI: 1.07, 1.31), and low (OR = 4.45 95 % CI: 1.19, 16.57) and medium (OR = 7.42, 95 % CI: 1.77, 31.02) levels of education were predictors of functional outcome. At 6 months, only the performance of decompressive craniectomy (OR = 4.31, 95 % CI: 1.37, 13.58) and a greater length of stay on MVS (OR = 1.13, 95 % CI: 1.05, 1.21) were variables associated with a higher risk of a GOSE score ≤ 6. Finally, age > 65 years (OR = 4.96, 95 % CI: 1.12, 21.84), a greater length of stay on MVS (OR = 1.11, 95 % CI: 1.03, 1.13), and low (OR = 6.12, 95 % CI: 1.48, 25.16) and medium (OR = 6.49, 95 % CI: 1.49, 28.21) levels of education were associated with a higher risk of an unfavorable functional outcome 12 months after the trauma (Table 2).
Final logistic regression model for functional outcome at hospital discharge and 3, 6, and 12 months after TBI.
GOSE, Glasgow Outcome Scale-Extended; ISS, Injury Severity Score; LOS, length of stay; MVS, mechanical ventilation support.
This prospective study reveals a dynamic and evolving recovery process of individuals with severe TBI over the period of 1 year after the injury. Both at the time of hospital discharge and 3 months after the traumatic event, most patients showed significant levels of functional dependence, with approximately 20 % registering a high degree of disability. This high rate of disabilities in the acute phase is consistent with findings in other studies that evaluated short-term functional recovery.32–33 However, it can be observed that by reaching the 6-month mark, a significant portion of patients (38 %) showed a better outcome, with 19 % presenting moderate disability and another 19 % achieving complete recovery. This finding agrees with the results of previous studies with 6 months follow-up after sTBI,34–35 although it diverges from the findings of a previous study conducted in Brazil where functional recovery levels were higher in the first 3 months.18 As the follow-up extended to the 12-month period, there was a notable increase in the number of individuals who achieved total or good recovery, indicating that, over time, a substantial portion of patients with severe TBI showed significant improvement in their functionality and independence and those results are in line with previous studies that have conducted long-term follow-ups.36–37
Age above 65 was a strong predictor of an unfavorable functional outcome 12 months after the traumatic event. Other studies found similar results, with an almost linear relationship between advanced age and functional outcome on the GOSE after 6 months,8,38 and 12 months postinjury.39 In line with the present study’s results, a previous study did not find an association between age and functional outcomes at 3 months.40 Moreover, despite most patients being male, sex was not a predictor of hospital mortality nor long-term functional recovery, only showing as a predictive factor for functional outcome at the time of hospital discharge. This finding supports previous findings where age and sex were not associated with functional recovery 24 months post-trauma,41 which suggests that in the long term, the level of independence of these patients may be explained by other factors besides demographic ones.42
Regarding trauma severity indices, ISS scores >25 were associated with a higher risk of unfavorable outcomes at 3 months post-TBI. Although trauma severity indices are widely used in predicting hospital outcomes, few studies have included this variable in the final regression model. Other clinical factors such as GCS score at admission,37 length of ICU stay,18 time to obey commands,39 and changes in cranial CT scans43 were reported in previous studies, suggesting they have greater predictive potential for functional outcomes after severe TBI.
The performance of decompressive craniectomy was a predictor of worse functional outcome at 3- and 6-months post-trauma. This finding diverges from results found in previous studies,44–45 where decompressive craniectomy in cases of acute subdural hematoma post-TBI increased the chances of a favorable functional outcome. Additionally, one study34 did not find an association between decompressive craniectomy and GOSE scores 6 months post-trauma. However, it is worth noting that only a few studies have evaluated decompressive craniectomy within a final prediction model of functional outcome.
Length of stay on MVS was a predictor of poorer functional recovery at all follow-up points. A study found similar relationships regarding GOSE scores 6 months after sTBI.8 However, it is worth noting that the average days on MVS in the present study sample were considerably higher than those found in other studies.46–47
Higher education levels were strong predictors of better functional recovery at 3 and 12 months. These findings are in line with previous studies39 where individuals with more favorable sociodemographic conditions had higher survival rates and better functional outcomes. Given that education is a modifiable factor, and considering that approximately 55 % of individuals in our sample have low education, this finding is consistent with previous studies from LMICs, which indicate that individuals with lower educational level are more likely to experience worse functional outcomes after TBI. However, as our data reflect the educational distribution of the broader population, we acknowledge that further population-based studies are needed to determine whether individuals with low education have a higher incidence of TBI compared to other educational strata.48
In the present study, approximately 50 % of individuals with severe TBI demonstrated a positive functional outcome 12 months after the traumatic event. However, it is crucial to emphasize that functional recovery showed a progressive, but not resolving trend, as the prevalence of disabilities and dependence was still considerable. These results align with findings from previous studies,9,19,49–52 which indicate that patients continued to face functional limitations and restrictions in social participation even over a period of 10 years post-traumatic brain injury.
LimitationsThis study has limitations that should be acknowledged. Firstly, the single-center setting may limit the generalizability of the findings to other populations or healthcare settings. Secondly, the lack of data on computed tomography (CT) scans represents a significant limitation, as CT results are strong predictors of outcomes after TBI. Due to the absence of a researcher trained in interpreting these exams, CT data could not be included in our analysis. Additionally, unmeasured covariates, such as whether patients received rehabilitation after hospital discharge, may have influenced the results. This missing information could have provided valuable insights into the functional recovery process. Despite these limitations, this study presents trends in functional recovery after severe TBI in a low- to medium-income country. Most studies on long-term TBI outcomes come from high-income settings; therefore, these findings help trace the functional trajectory from hospital admission to one-year post-injury and may aid in understanding recovery patterns in patients with disadvantaged social conditions in high-risk environments for severe TBI.
ConclusionsAge, high values in the Injury Severity Score, length of stay in mechanical ventilation, the performance of decompressive craniectomy, and level of education were independent variables associated with functional outcome over the first year after severe TBI. The results of the present study demonstrate the influence of clinical and sociodemographic parameters on mortality and functional recovery outcomes after severe TBI. It provides a comprehensive view of the functional trajectory from hospital admission to one year after the traumatic event. The study highlights the need for further research with larger sample sizes, multicenter settings, and longer follow-up periods to track the functional trajectory.
AcknowledgementsThe present study was funded by Fundação de Amparo à Pesquisa (FAPES), Grant ID 09/2020. We acknowledge the study participants and the directors of the Hospital Estadual de Urgência e Emergência for their support.
The authors declare no competing interest.