There is about ten years since the first randomized controlled trial looking for the effects of photobiomodulation therapy using low-level laser therapy and/or light emitting diodes therapy in athletic performance enhancement was published. Since then, the knowledge in this field has increasing exponentially.
ObjectiveGiven the fast advance in clinical interest, research and development in the use of photobiomodulation therapy for athletic performance enhancement and also to accelerate post-exercise recovery, as pioneers in this research field we felt the need to establish recommendations to ensure the correct use of the therapy, and also to guide the further studies in this area looking for the achievement of highest scientific evidence. It is important to highlight that the establishment of both clinical and scientific recommendations in this masterclass article were based on the most recent systematic reviews with meta-analysis and randomized controlled trials published in this field. It is important to stress that the recommendations of this masterclass article are based on most recent systematic reviews with meta-analysis and RCTs published in this research field. Future guidelines must follow the same direction and must be based only at the highest scientific evidence, avoiding overstatements and extrapolations based on animal experiments and case-studies.
Ten years ago, in 2008, the first randomized controlled trial (RCT) investigating the use of photobiomodulation therapy (PBMT) for athletic performance enhancement was published1 only two years after the first animal study in this field be published.2 This pioneer RCT1 showed for the very first time that the treatment with PBMT before an exercise session could enhance the performance of high-level volleyball athletes, decrease the delayed onset muscle fatigue, and prevent the expected increase of blood lactate levels.
In the next two years a series of RCTs involving elite athletes was published by our group investigating different PBMT parameters as well as different functional and biochemical markers.3–8 The outcomes of these RCTs confirmed the potential of PBMT as an ergogenic agent, and as a therapeutic tool to enhance post-exercise recovery. After the initial skepticism started to decrease, then other research groups started to develop and publish studies in the same area.9–12 Their results mostly confirmed what was previously observed, and the knowledge in this field started to increase exponentially.
At the year of 2013, the first meta-analysis in this field was published also by our research group13 (ahead of print version), and at that time 13 RCTs with acceptable methodological quality were included and the outcomes showed that PBMT was able to increase the number of repetitions and the time to exhaustion if applied before an exercise session. Four years later the most recent meta-analysis was published14 now including 39 RCTs, and for the very first time it was possible to clearly identify a therapeutic dose window for large and small muscle groups. This increasing evidence lead to the International Olympics Committee to recommend PBMT as a therapeutic agent to improve acute muscular recovery,15 which is an unquestionable milestone to this field.
Currently, only one year after the publication of the most recent meta-analysis,14 we can identify more than 50 RCTs published in this field showing that PBMT can not only increase exercise performance in healthy subjects in a laboratory-controlled environment, but also in high-level athletes in field tests16 and in real sports settings,17 and in patients with different medical conditions such as chronic obstructive pulmonary disease (COPD),18,19 fibromyalgia,20 and chronic kidney disease.21 This fast growing has also attracted the industry's interest, leading to the development of devices designed specifically for exercise performance enhancement.
Given the fast advance in clinical interest, research and development in the use of PBMT for athletic performance enhancement and also to accelerate post-exercise recovery, we felt the need to establish a guideline to ensure the correct use of the therapy, and also to guide the further studies in this area looking for the achievement of highest scientific evidence. It is important to highlight that the establishment of both clinical and scientific guidelines below were based on the most recent systematic review and meta-analysis published in this field,14,22 and we considered also the RCTs published after the publication of this systematic review (but following the same criteria used in the most recent systematic review), up to November 2018.
Clinical recommendationsPBMT, also known as phototherapy, is a nonthermal process where light interacts with chromophores leading to photophysical and photochemical reactions in different tissues. PBMT is a light therapy that uses non-ionizing light sources, such as lasers, light emitting diodes (LEDs), and broadband light, from the visible to the infrared spectrum.
As mentioned before, the following parameters described in this guideline were extracted from the most recent systematic reviews with meta-analysis in this field,14,22 and also from the RCTs with high methodological quality published after the aforementioned reviews, up to November 2018. These recommendations are for young/adult and healthy subjects, since currently there is not enough studies to support recommendations for non-healthy or elderly subjects.
- •
Light source: Lasers and/or LEDs;
- •
Dose: 20 to 60J dose for small muscle groups (i.e. biceps brachii or triceps surae), 60 to 300J for large muscle groups (i.e. quadriceps and hamstrings);
- •
Power: 50 to 200mW per diode (for single probes); 10 to 35mW per diode (for cluster probes). The total power of the device can not lead to thermal effects;
- •
Wavelengths: 640nm (red) to 950nm (infrared). More recently, most studies published in this field have combined red and infrared wavelengths simultaneously;
- •
Mode: Pulsed or continuous;
- •
When to irradiate: For acute effects (a single event) 5min to 6h before activity.16,17,23 For chronic effects associated to strength training the irradiation must be performed immediately before each exercise session (5 to 10min).24 For chronic effects associated to endurance training (treadmill) the irradiation must be performed immediately before and immediately after each exercise session (5 to 10min)25;
- •
Length of irradiation: minimum of 30s per site/point;
- •
Where to irradiate: The irradiation must cover as much of the area as possible in most (preferably all) muscle groups involved in exercise activity. If single probes are being used, the distance between irradiation sites/points must be lower than 2cm;
- •
Irradiation technique: Direct contact with skin (stationary) with slight pressure.
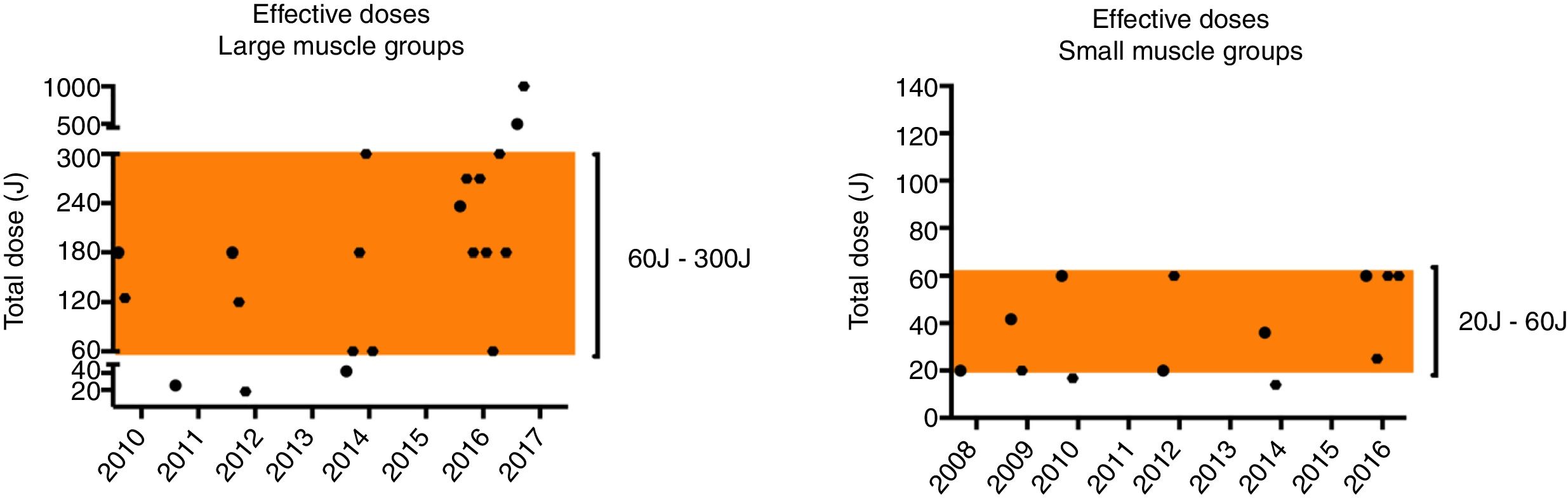
A special highlight must be given regarding the current dose range established in the most recent systematic review.14 It was evidenced that 75% of doses that presented positive results for performance enhancement in large muscle groups were between 60J and 300J, and that 85% of doses that presented positive results for performance enhancement in small muscle groups were between 20J and 60J (Fig. 1).
Current therapeutic window based on most recent systematic review14 in the research field.
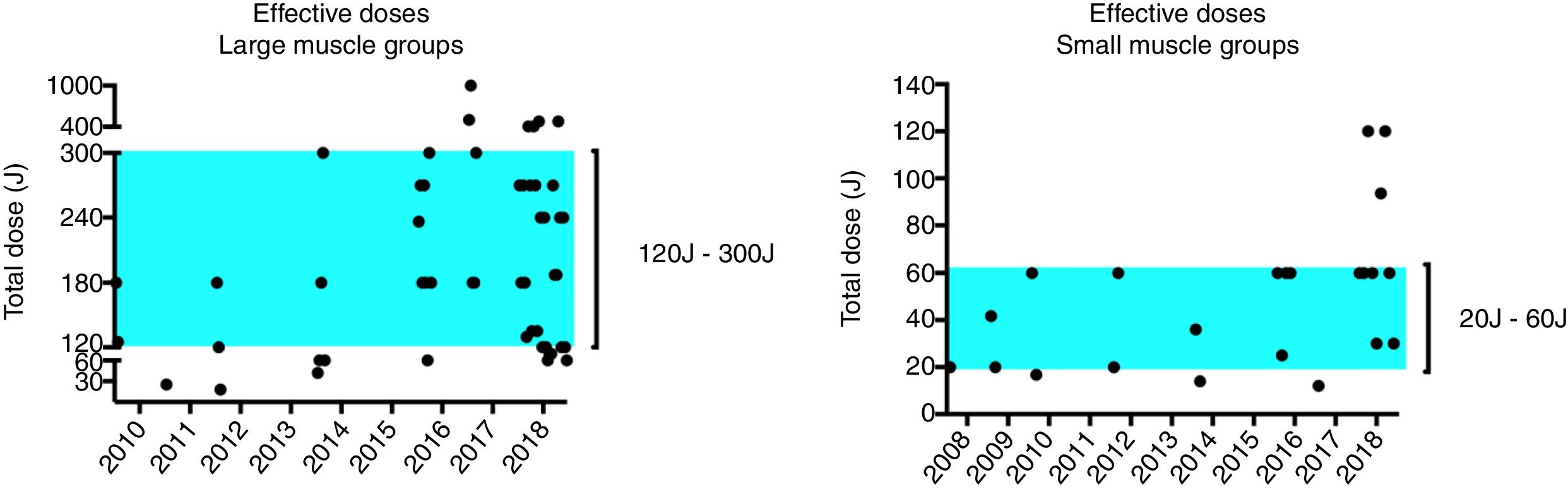
However, as mentioned previously, since the publication of the most recent systematic review14 we can identify the publication of more 15 RCTs, up to November 2018.17,23,25–37 Taking into consideration the doses that presented positive outcomes in these RCTs,17,23,25–35,37 we were able to identify that currently around 70% of doses that presented positive results for performance enhancement in large muscle groups are between 120J and 300J, and that around 77% of doses that presented positive results for performance enhancement in small muscle groups stay between 20J and 60J (Fig. 2). Interestingly, among all of the fifteen recent published studies above mentioned17,23,25–37 that used doses inside the proposed 120J to 300J dose therapeutic window for large muscle groups, there is only one36 that did not get positive outcomes for performance enhancement. Moreover, in the same study36 a high dose of 180J was used for calf muscles, which is 3 times higher than the 60J dose threshold recommended for small muscle groups.
Updated therapeutic window considering the 15 RCTs17,23,25–37 published after the systematic review,14 up to November 2018.
It is important to highlight also that in studies comparing doses inside the 120J to 300J therapeutic window against doses above 300J for large muscle groups (405J and 480J for instance), the doses inside the 120J to 300J therapeutic window presented better outcomes.
Scientific recommendationsDespite the growing body of evidence supporting the use of PBMT for performance enhancement and to accelerate post-exercise recovery, the quality of evidence needs to be improved. The recent systematic reviews with meta-analysis in this field14,22 showed low to moderate quality of evidence to the main outcomes, demonstrating that further research is needed to be confident about the effects. We attribute this quality level of evidence mainly to the risk of bias category and the imprecision of the results due the small sample size and wide confidence intervals observed in the outcomes.
We strongly recommend the attention of researchers to perform the prospective register of study protocol (and to publish the protocol in peer-reviewed journals when possible),38,39 to follow the Consolidated Standards of Reporting Trials (CONSORT) guidelines when designing the trials, and the use of the TIDieR checklist (template for intervention description and replication) to confirm if all items required were reported in the manuscript before submission.40,41 This is a way to reduce reporting bias and assist the authors to follow an adequate, clear, and transparent reporting of data and study design.
A special attention must be paid for the report of crossover trials since there is not a specific guideline for it. Furthermore, future studies should present their data in absolute values and their respective variation, as mean and standard deviation (± SD), with detailed description besides percentages, change in outcomes, etc. Further concern should be taken in reporting PBMT parameters. These parameters should be shown in detailed form, such as in a table in the manuscript, to provide more information for the reader regarding the features of the device used and allow the study replication by other authors. Also, it is important to state if the parameters used match or not to the current scientific evidence in order to advance on it.
Final considerationsIt is important to stress that these recommendations were based on most recent systematic reviews with meta-analysis and RCTs published in this research field. Future guidelines must follow the same direction and must be based only at the highest scientific evidence, avoiding overstatements and extrapolations based on animal experiments and case-studies for instance.
Conflicts of interestProfessor Ernesto Cesar Pinto Leal-Junior receives research support from Multi Radiance Medical (Solon, OH), a laser device manufacturer. Multi Radiance Medical had no role in the planning of this masterclass article, and the statements are not based in their devices. The remaining authors declare that they have no conflict of interests.