Osteoporotic vertebral fractures affect a large number of older adults
ObjectivesSystematically review evidence of the benefits and harms of non-surgical and non-pharmacological management of people with osteoporotic vertebral fractures compared with standard care (control); and evaluate the benefits and harms of non-surgical and non-pharmacological management of people with osteoporotic vertebral fractures compared with an alternative non-pharmacological, non-invasive intervention.
DesignSystematic review and meta-analysis of randomized controlled trials. Five electronic databases (CINAHL, EMBASE, MEDLINE, PUBMED, and COCHRANE) were searched. Eligible trials included participants with primary osteoporosis and at least one vertebral fracture diagnosed on radiographs, with treatment that was non-surgical and non-pharmacological involving more than one session.
ResultsTwenty randomized controlled trials were included with 2083 participants with osteoporotic vertebral fractures. Exercise, bracing, multimodal therapy, electrotherapy, and taping were investigated interventions. Meta-analyses provided low certainty evidence that exercise interventions compared to no exercise were effective in reducing pain in patients with osteoporotic vertebral fractures (mean difference (MD)= 1.01; 95% confidence interval (CI): 0.08, 1.93), and low certainty evidence that rigid bracing intervention compared with no bracing was effective in reducing pain in patients with osteoporotic vertebral fractures (MD= 2.61; 95%CI: 0.95, 4.27). Meta-analyses showed no differences in harms between exercise and no exercise groups. No health-related quality of life or activity improvements were demonstrated for exercise interventions, bracing, electrotherapy, or multimodal interventions.
ConclusionsExercise and rigid bracing as management for patients with osteoporotic vertebral fractures may have a small benefit for pain without increasing risk of harm.
Trial registrationPROSPERO registration number CRD42012002936
Vertebral fractures are a common consequence of osteoporosis. Annually, there are 400,000 osteoporotic vertebral fractures (OPVFs) occurring in the European Union 6 (United Kingdom, Italy, France, Germany, Spain, plus Sweden)1 and this number is expected to increase by 23% by the year 2030.1 Typically, OPVFs result in significant and persistent back pain, spinal deformity, and reduced mobility and quality of life.2-4 OPVFs also are a marker of poor bone health and predict future fractures, both vertebral and non-vertebral.5-6 Lindsay et al5 reported that a single vertebral fracture is associated with a five-fold increase in further vertebral fractures. OPVFs are also a burden to the community with the mean cost of care for the 12-month period following an OPVF ranging from €1928 to €14,474 (approximately 13,156 BRL to 98,763 BRL).1
Surgical and pharmacological interventions commonly used for the management of OPVFs have limited efficacy and potential risk of harm. A recent Cochrane review7 reported moderate to high certainty evidence that vertebral augmentation, a percutaneous injection of bone cement under image guidance into a fractured vertebra for patients with OPVFs (acute or subacute), showed no important change for pain, quality of life, or disability when compared to sham treatments. The authors reported uncertainty regarding risk of this procedure, including risk of future fracture. Another systematic review showed very low certainty evidence that opioids and non-steroidal anti-inflammatory medications provide acute pain relief for people with OPVFs when compared to alternative medicine.8 Concerningly, opioids showed no significant effects for pain relief when compared to placebo, and were associated with more adverse events such as gastrointestinal disorders.8
In comparison to vertebral augmentation and analgesia or anti-inflammatory medication, non-surgical and non-pharmacological management is non-invasive and might be expected to result in less harm. Further, these interventions (e.g. exercise therapy, bracing) may address problems associated with OPVFs including pain and limited activity. Previous systematic reviews have shown very low certainty evidence that exercise interventions for people with OPVFs7-9 may have small benefits for pain, quality of life and physical performance and few adverse events. Rzewuska et al's10 systematic review demonstrated low certainty evidence for the use of spinal orthoses for managing pain and disability in patients with OPVFs in the medium-term and very low certainty evidence for the effectiveness of a rigid orthosis compared to a soft spinal orthosis in reducing pain in the short-term.
Currently, there is insufficient evidence about the benefits and harms of non-surgical and non-pharmacological interventions to guide clinicians, which may lead to variable and suboptimal care for patients with OPVFs. Previous systematic reviews have not been comprehensive in their inclusion of a range of non-surgical and non-pharmacological interventions for OPVFs and little emphasis has been given on synthesising the evidence on harms as well as benefits.7-10 As literature searches in other reviews have not been updated since 2017 there is a need to consider new randomized controlled trials completed since.
This systematic review aims to evaluate the evidence from randomized controlled trials on the benefits and harms of non-surgical and non-pharmacological management of people with OPVFs compared with standard care (control); and, to compare the benefits and harms of non-surgical and non-pharmacological management of people with OPVFs compared with an alternative non-pharmacological, non-invasive intervention.
MethodsA systematic review and meta analyses were conducted following the PRISMA Statement.11 This systematic review was prospectively registered on the PROSPERO database (CRD42012002936).
Eligibility criteriaThe trials were eligible if they were: randomized controlled trials; included participants with a diagnosis of primary osteoporosis and a vertebral fracture demonstrated on radiographs; the experimental group received non-pharmacological or non-surgical management of more than one session (e.g., strengthening, balance, or motor control exercise interventions, bracing, taping, electrotherapy) and the comparison group received standard care or an alternative intervention (e.g., soft brace compared to rigid brace); and if at least one outcome was reported at the conclusion of the intervention.
Search strategyThe electronic databases MEDLINE, EMBASE, CINAHL, PUBMED, and COCHRANE were searched until April 2021. Search strategies for each database were constructed using the concepts of population, intervention, and design. Population was defined as participants with primary osteoporosis and at least one vertebral fracture demonstrated on radiographs. Intervention was defined as a non-surgical or non-pharmacological intervention for OPVFs. The design was limited to randomized controlled trials. Within each concept, synonyms and MeSH terms were combined with the “OR” operator. The concepts were then combined with the “AND” operator (see Supplemental online material). Database searching was supplemented by checking reference lists of included trials and citation tracking.
Study selectionThe first 50 titles and abstract were assessed by two reviewers independently to pilot inclusion and exclusion criteria. After confirming high levels of agreement, all titles and abstracts were assessed independently by two reviewers according to the eligibility criteria. Full copies of articles unable to be excluded by this process were obtained and again reviewed. Disagreements were resolved by discussion, and in one case a third reviewer was consulted to achieve consensus.
Data extractionThe following data were extracted: author, year and country of publication, characteristics of participants (sex, age); description of intervention and control; pain measures (visual analogue scale), activity measures, quality of life measures, adverse events and participant adherence to interventions, type of comparison, and studies included in the analysis. Data extraction was performed by one reviewer and checked by a second reviewer.
Risk of biasThe methodological quality of the trials was assessed by two reviewers independently using the Physiotherapy Evidence Database (PEDro). Any differences in assessment were resolved by discussion. The PEDro score has reported inter-rater reliability of moderate range [ICC= 0.68 (95%CI 0.57, 0.76)]12 and the use of a summed score out of 10 has been confirmed using Rasch analysis.13 Trials with a score of 6 or more were considered to be of high quality.14
Data analysisThe primary outcome of benefit was determined by the effect of the intervention on pain reduction, and the secondary outcome of benefit was determined by the effect of the intervention on activity and health-related quality of life. The primary outcome of harm was determined by the number of participants experiencing serious and non-serious adverse events. A serious adverse event was defined as the number of participants experiencing an event that was fatal, life threatening, requiring hospitalisation, and/or fractures, or as reported in the included trial as a serious event. A non-serious event was defined as any other adverse event.
For continuous outcomes (pain, activity, quality of life) the post-intervention means ± standard deviation (SD) were extracted. If a mean change score was reported, the post-intervention mean was calculated by adding the mean change to the baseline mean; and baseline SD was used as an estimate of the post intervention SD. Pain outcomes for individual trials were transformed to a common scale (0 to 10). For dichotomous outcomes (adverse events) the number of participants experiencing an adverse event or serious adverse event was recorded.
Meta-analysis was completed using a random effects model and expressed as mean differences (MD) for pain or standardised mean differences (SMD) for activity and quality of life, and relative risks for adverse events. Data were combined for meta-analysis using a minimum of two trials assessed as clinically homogeneous. Trials were considered clinically homogenous if there was a common intervention and outcome. Trial populations were homogenous based on the eligibility criteria. Data used for analysis were taken from assessments at completion of intervention. All analyses were conducted in Review Manager (RevMan) software (Version 5.3.5).15 The results from the trials not able to be combined into meta-analyses were reported in table and narrative form. Positive SMD values were used to indicate that the outcome was favourable to the intervention group. SMD values of < 0.2 indicated a small effect size, 0.2-0.5 a moderate effect size and > 0.8 a larger effect size.16
The GRADE approach was applied to each meta-analysis to assess certainty of evidence.17 This process involved downgrading from high to moderate to low to very low certainty evidence based on domains. Downgrading the evidence one level if: (1) for study limitations, the PEDro score was <6 for the majority (> 50%) of trials in the meta-analysis, (2) for inconsistency, there was greater than low levels of statistical heterogeneity between trials (I2≥ 25%),18 (3) for indirectness, more than 50% of the participants were outside the target group, (4) for imprecision, there were large confidence intervals, such that the lower band of the confidence interval of the estimate of effect could indicate little or no effect, while the upper band could indicate a large effect, (5) for publication bias, as indicated by funnel plot asymmetry on any meta-analysis with at least 10 trials.18 The certainty of evidence was downgraded more than one level based within one domain if there were very serious concerns (for example, for study limitations if the PEDro score was <6 for >75% of trials), or if there were serious concerns in more than one domain. Single randomized trials were considered inconsistent and imprecise, thereby providing low certainty evidence. This could be further downgraded to very low certainty evidence if there was also high risk of bias.19 The GRADE domains were applied by one researcher and checked by a second researcher.
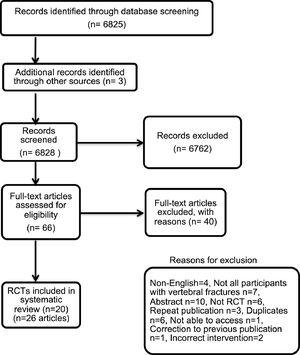
ResultsStudy selectionThe database search yielded a total of 6,348 articles including duplicates. Forty-five articles were included for full text review and a total of 20 trials (reported in 26 articles) met the inclusion criteria (Fig. 1).
Study characteristicsA total of 20 randomized controlled trials including 2,083 participants were included. The majority of participants were women (1,873, 89.9%). Participants were predominantly community dwelling, with a mean (SD) age of 72.8 ± 6.2 years. All trials were completed in countries classified as high or upper-middle income status (Table 1).
Trial characteristics.*
| Trial (Economy classification:High, upper-middle, middle, low)51 | Participants (intervention)Number, NMean ± SD ageSex, n (%) | Participants (comparator)Number, NMean ± SD ageSex, n (%) | Setting | Intervention components | Dosage | Length of intervention | Comparator |
|---|---|---|---|---|---|---|---|
| Barker et al 201920 (UK)(High) | N = 21672.2 ± 8.4 yearsFemale,185 (85.6%) | N = 19672.2 ± 8.4 yearsFemale, 173 (88.7%) | Clinic and home program | Exercise | Strength training intensity set at RPE level 3-445min session/week,3-5/week | 6 sessions over 12 weeksHome program for 12-months | No exercise |
| Bergland et al 201129(Norway)(High) | N = 4770.8 ± 5.9 yearsFemale, 47 (100%) | N = 4272.0 ± 5.8 yearsFemale, 42 (100%) | Clinic | Exercise | Biweekly 3-month program | No exercise | |
| Berstrom et al 200121(Sweden)(High) | N = 2073.2 ± 8.9 yearsFemale, 20 (100%) | N = 1674.1 ± 6.0 yearsFemale, 16 (100%) | Clinic | Exercise - strengtheningPostural advice | x30 progressive increase resistance | Biweekly 4-month program | Not stated |
| Cergerl et al 201923(Turkey)(Upper-middle) | Supervised exercise N = 2058.9 ± 4.7 yearsFemale, 20 (100%) | N = 2059.65 ± 6.45 yearsFemale, 20 (100%) | Clinic or home program | Exercise | 3 × 8 reps for first 2 weeks.x3 × 10 reps, progress to x3 × 12 reps | 6 weeks | |
| Evstigneeva et al 201622(Canada)(High) | N = 4070.7 ± 8.1 yearsFemale, 40 (100%) | N = 3867.6 ± 7.0 yearsFemale, 38 (100%) | Clinic | Exercise | 40-minute program twice weekly | 12 months | No exercise |
| Giangregorio et al 201827(AustraliaCanada)(High) | N = 7176 ± 6.4 yearsFemale,71 (100%) | N = 7077 ± 7.3 yearsFemale,70 (100%) | Home programSupported by 6 home visits by physical therapist | Exercise | 5-8 exercises2 sets of 8-10 reps** or isometric holds of 3-5 seconds | 12 months | No exercise |
| Gold et al 200426(USA)(High) | N = 9480.2 ± 4.8 yearsFemale, 94 (100%) | N = 9182.0 ± 6.2 yearsFemale, 91 (100%) | Clinic | ExerciseEducation | Exercise 3x week for 6 monthsCoping class x2 week for 6 months | 6 months, cross over with control | No exercise |
| Grahn et al 202032(Sweden)(High) | N = 1071.3 ± 5.3 yearsFemale, 9 (90%) | N = 1072.4 ± 6.5 yearsFemale, 10 (100%) | Fitness Centre and home exercise program | Yoga/mindfulness | Hour class weekly: 30 min yoga/30 min mindfulness trainingDaily home exercise 30-minutes duration | 10 weeks | No exercise |
| Karaksidou et al 201325(Greece)(High) | N = 1067.6 ± 6.41 yearsFemale, 10 (100%) | N = 1069.3 ± 4.4 yearsFemale, 10 (100%) | Clinic | Exercise | x3 week 1-hour one-to-one sessions with physical therapist | 13-week program | No exercise |
| Malmros et al 199824(Denmark)(High) | N = 2765 years(25:75 percentile 62:70) | N = 2568 years(25:75 percentile 64:71) | Clinic and home exercise program | Exercise | 60-minute twice weekly physical therapy-led sessions | 10-week program | No exercise |
| Papaioannou et al 200228(Canada)(High) | N = 3771.6 ± 7.3 yearsFemale, 37 (100%) | N = 3772.2 ± 7.98 yearsFemale, 37 (100%) | Home program | Exercise | 60-minutes x3 per week | 12 months | No exercise |
| Stranghelle et al (2019)30(Norway)(High) | N = 7674.6 ± 6.1 yearsFemale, 76 (100%) | N = 7373.7 ± 5.6 yearsFemale, 73, (100%) | Clinic | Exercise | One session, twice weekly, physical therapist led sessions | 12 weeks | No exercise |
| Kato et al 201937(Japan)(High) | N = 14176 ± 5.2 years | N = 14375.5 ± 5.4 years | Brace - rigid | Wore brace at all times | 12 weeks | Soft brace | |
| Kim et al 201936(Korea)(High) | Soft braceN =2066.8 ± 11.0 yearsFemale, 14 (70%)Rigid braceN =2071.8 ± 8.0 yearsFemale, 14 (70%) | N = 2072.2 ± 10.4Female, 13 (65%) | Brace - rigid or soft | Worn at all times except when lying down | 8 weeks | No brace | |
| Li et al 201535Hong Kong(High) | N = 2782 ± 8.3 yearsFemale, 27 (100%) | N = 2481 ± 6.6 yearsFemale, 24 (100%) | Brace – rigid or Spinomed | Week 1:24 hours per dayWeeks 2-3: 3 hours daily | 3 weeks | Soft lumbar orthosis | |
| Pfeifer et al 201134(Germany)(High) | Group A N = 3672.8 ± 7.1 yearsFemale, 36 (100%)Group B N = 3672.3 ± 6.7 yearsFemale, 36 (100%) | N = 3669.7 ± 8.9 yearsFemale, 36 (100%) | Brace – Rigid or soft | 2 hours daily | 6 months | No brace | |
| Barker et al 201943 (UK)(High) | N = 203 72.4 ± 9.3 yearsFemale, 173 (85.6%) | N = 19672.2 ± 8.4 yearsFemale, 173 (88.7%) | Clinic and home program | Multimodal therapy | 6 sessions over 12 weeksHome program for 12-months | 12 months | No therapy |
| Bennell et al 201040(Australia)(High) | N = 1166.2 ± 8.0 yearsFemale, 7 (63.6%) | N = 966.3 ± 11.8 yearsFemale 7 (88.7%) | Clinic and home program | Multimodal therapy | 45-minute sessionStandardised treatment, with individualised dosage | 10 weekly sessions | No intervention |
| Palmer et al 201844(UK)(High) | N = 1373.6 ± 5.9 yearsFemale, 13 (100%) | N = 1274.6 ± 7.0 yearsFemale, 8 (67%) | Taping spine | Daily length of time of application chosen by patient | 4 weeks | Usual care | |
| Rossini et al 200938(Italy)(High) | N = N = 2073.8 ± 7.4 yearsFemale, 20 (100%) | N = 2171.7 ± 7.2 yearsFemale, 21 (100%) | Home program | Electrotherapy | 10 hours per day | 2 months | Sham therapy |
| Zambito et al 200739(Italy)(High) | Group 1N =3570.8 ± 7.4 yearsFemale, 35 (100%)Group 2 N =3570.5 ± 7.6 yearsFemale, 35 (100%) | N = 3570.5 ± 8.3 yearsFemale, 35 (100%) | Clinic | Group 1Electrotherapy interferential therapy, trunk flexion-extension stretchingGroup 2Electrotherapy - horizontal therapy, trunk flexion-extension stretching | 30-40min, 5 days per week | 2 weeks | Sham horizontal therapy |
There were four categories of intervention: exercise,20-33 bracing,34-37 electrotherapy,38-39 and multimodal therapy including manual therapy, postural advice, exercise, and taping.20,40
Adherence to exercise interventions conducted in clinic settings ranged from “fully compliant” 23-25 to a mean attendance of 58% of sessions26 (Supplemental online material). Adherence to exercise interventions conducted in home settings (unsupervised) ranged between 62% to 66% of prescribed sessions in trials that reported data27-28 and undefined in one trial.23 Exercise trials with longer intervention periods ranging from 6 to 12 months exhibited lower adherence rates compared to trials with shorter intervention periods. For example, in one study 66% of participants attended 60% of scheduled exercise classes, with an observed reduction in this rate over the 12-month intervention.27 In comparison, an adherence rate of 100% was reported for two trials with shorter interventions of six and 10 weeks.23-24 Adherence to electrotherapy interventions was high with 100% of sessions attended39 and 9.2 hours received of the prescribed 10 hours of electrical stimulation per day.38 Reporting of adherence for bracing trials was highly variable, ranging from no data provided34-35 to 64% of participants wearing a rigid brace and 63% of participants wearing a soft brace for >12 hours at 3-month follow-up.37
Risk of bias in individual studiesSixteen trials were ranked as higher quality (≥6/10). Most adhered items on the PEDro scale included eligibility criteria, random allocation, and provision of both point measures and measures of variability for at least one key outcome. Eleven trials did not fulfil the criterion of concealed allocation increasing risk of selection bias, and none of the trials blinded the participants or therapists, therefore increasing the risk of performance bias. Six trials reported that more than 15% of participants withdrew from the study, increasing the risk of attrition bias. One individual electrotherapy trial39 included a sham treatment group; however the authors did not explicitly describe blinding of the participants (Supplemental online material).
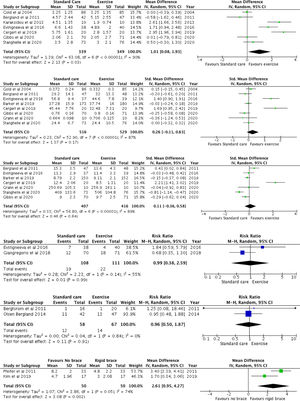
Synthesis of the resultsEffect of exercise interventions versus no exercise for the management of OPVFMeta-analyses of seven trials22-23,25-26,29-31 with 688 participants provided low certainty evidence that exercise interventions compared with no exercise were effective in reducing the primary outcome of pain in patients with OPVFs (MD = 1.01 units, 95% CI: 0.08,1.93)(Fig. 2A) (Table 2). Due to a high I2 value (90%), a sensitivity analysis compared exercise and no exercise for six trials with a small increase in effect size (MD = 1.25 units, 95% CI: 0.23, 2.27) but I2 remained high. This analysis was performed by omitting one trial29 as the exercise intervention focussed on balance exercise in comparison to strengthening interventions in the other six trials.
(A) Mean difference (95% CI) of effect of exercise vs. standard care on pain (B) Standardized mean difference (95% CI) of effect of exercise versus standard care on quality of life (C) Standardized mean difference (95% CI) of effect of exercise versus standard care on activity (D) Risk ratio (95% CI) of serious adverse events for exercise versus standard care (E) Risk ratio (95% CI) for non-serious adverse events for exercise versus standard care (F) Mean difference (95% CI) of effect of rigid brace versus no brace on pain.
Summary of findings and certainty of evidence GRADE for Exercise versus no exercise for OPVF.
| Certainty of Evidence | No. of Participants | Effect (95% CI) | Certainty | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of Studies | Study design | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication bias | Outcome | Exercise | Control | ||
| 7 | RCT | Not serious | Serious | Not serious | Serious | Undetected | Pain intensity | 349 | 339 | MD = 1.01(0.08, 1.93) | Lowab |
| 8 | RCT | Not serious | Serious | Not serious | Serious | Undetected | QOL | 529 | 516 | SMD = 0.26(-0.11, 0.63) | Lowab |
| 7 | RCT | Not serious | Serious | Not serious | Serious | Undetected | Activity | 416 | 407 | SMD = 0.11(-0.36, 0.58) | Lowab |
| 2 | RCT | Not serious | Serious | Not serious | Serious | Undetected | SAE | 111 | 108 | RR = 0.99(0.38, 2.59) | Lowac |
| 2 | RCT | Not serious | Not serious | Not serious | Serious | Undetected | NSAE | 67 | 58 | RR = 0.96(0.55, 1.87) | Moderatec |
MD, mean difference; NSAE, non-serious adverse event; QOL, quality of life; RCT, randomized controlled trial; RR, relative risk; SAE, serious adverse event; SMD, standardized mean difference.
Downgraded due to inconsistency there was statistical heterogeneity(I2 values > 25%) with the studies showing differing estimates of the effect size.
Meta-analysis of two trials22,27 with 219 participants provided low certainty evidence of no difference between exercise and no exercise groups regarding the primary outcome of risk of serious adverse events (RR = 0.99, 95%CI 0.38, 2.59) (Fig. 2E). Meta-analysis of two trials21,33 with 125 participants showed moderate certainty evidence of no difference between exercise and no exercise groups regarding risk of non-serious adverse events (RR = 0.96, 95%CI: 0.5, 1.87) (Fig. 2F). Eleven exercise trials20-22,24-28,30,33,40 with 1460 participants, reported adverse events ranging from two events25 to 184 events.27 One trial26 reported serious adverse events of two fractures related to the exercise intervention. Seven trials reported non-serious adverse events related to the interventions such as knee pain, neck pain, adverse skin reactions to tape, breathing difficulties.20,22,25-27,33,40
Meta-analyses provided low certainty evidence of no differences between exercise and no exercise regarding the secondary outcomes of health-related quality of life and activity (Fig. 2B and C) (Table 2). Other individual trials could not be included, as they did not assess these outcomes.21-22,27 Two trials involving a total of 553 participants provided low certainty evidence of similar costs and health utilisation for participants in exercise and comparison groups.41-42
Effect of bracing versus no bracing for the management of OPVFsMeta-analyses of two trials34,36 with 100 participants provided low certainty evidence that rigid bracing interventions compared with no bracing were effective in reducing the primary outcome of pain in patients with OPVFs (MD = 2.61 units, 95%CI: 0.95, 4.27) (Figure 2 F) (Table 3).
Summary of findings and certainty of evidence GRADE for Bracing versus no brace for OPVF.
| Certainty of Evidence | No. of Participants | Effect (95%CI) | Certainty | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of Studies | Study design | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication bias | Outcome | Brace | No brace | ||
| Rigid brace vs no brace | |||||||||||
| 2 | RCT | Not serious | Serious | Not serious | Serious | Undetected | Pain intensity | 50 | 50 | MD = 2.61(0.95, 4.27) | Lowab |
| Soft brace vs no brace | |||||||||||
| 2 | RCT | Not serious | Serious | Not serious | Serious | Undetected | Pain intensity | 49 | 50 | MD = 2.39(-0.45, 5.23) | Lowab |
| Rigid brace vs no brace | |||||||||||
| 2 | RCT | Not serious | Serious | Not serious | Serious | Undetected | QOL | 50 | 50 | SMD = -0.55(-1.43, 0.33) | Lowab |
| Soft brace vs no brace | |||||||||||
| 2 | RCT | Not serious | Serious | Not serious | Serious | Undetected | QOL | 49 | 50 | SMD = -0.49(-1.76, 0.78) | Lowab |
| Rigid brace vs no brace | |||||||||||
| 2 | RCT | Not serious | Not serious | Not serious | Serious | Undetected | Activity | 50 | 50 | SMD = -0.13(-0.54, 0.28) | Moderateb |
| Soft brace vs no brace | |||||||||||
| 2 | RCT | Not serious | Serious | Not serious | Serious | Undetected | Activity | 49 | 50 | SMD = 0.68(-0.84, 2.19) | Lowab |
MD, mean difference; QOL, quality of life; RCT, randomized controlled trial; SMD, standardized mean difference.
Three bracing trials with 452 participants reported adverse events.34,36-37 Serious adverse events included vertebral fractures (n = 16, no significant difference rigid versus soft braces),37 and death unrelated to intervention.36 Non-serious adverse events included pain and discomfort (n = 5).34
Meta-analyses provided low to moderate certainty evidence of no differences between rigid bracing interventions compared with no intervention regarding the secondary outcomes of quality of life or activity (Table 3). Meta-analysis of trials34,36 investigating soft bracing compared with no bracing provided low certainty evidence of no differences in reducing pain, improving quality of life, or activity. An individual trial could not be included, as it did not assess these outcomes, or provided sufficient data.37
Effect of rigid bracing versus soft bracing for the management of OPVFsMeta-analyses of four trials34-37 with 413 participants provided moderate certainty evidence that rigid bracing in comparison to soft bracing interventions showed no difference in pain (MD = -0.10 units, 95%CI: -0.55, 0.35), and low certainty evidence of no difference in activity level (SMD = 0.19, 95%CI: -0.20, 0.58). Meta-analyses of three trials34,36-37 with 362 participants provided moderate certainty evidence that rigid bracing in comparison to soft bracing interventions showed no difference in quality of life (SMD = -0.09, 95%CI: -0.30, 0.11) in patients with OPVFs.
Effect of electrotherapy versus control (sham) for the management of OPVFsIndividual trials could not be combined in meta-analyses for electrotherapy due to unavailable data and clinical heterogeneity. One trial38 with 41 participants provided very low certainty evidence of no difference in quality of life measures (SMD = 0.27, 95% CI: -0.35, 0.89) compared to sham electrotherapy.
One trial38 reported nine adverse events, five occurred in the sham group and four in the intervention group.
Effect of multimodal therapy and taping versus standard care for the management of OPVFsOne trial40 of multimodal therapy comprising manual therapy, taping, and education compared to usual care with 20 participants demonstrated low certainty evidence of reduced pain at rest (MD = -2.0 units, 95% CI: -3.8, -0.2) in favour of the intervention group. Changes were also demonstrated for measures of quality of life and activity, in favour of the intervention group. One low quality trial43 of taping of the lumbar spine compared to standard care demonstrated non-significant findings for pain at rest (MD = 0.25, 95% CI: -1.75, 2.25) and quality of life (SMD = 0.67, 95% CI: -0.18, 1.47), and cutaneous reaction to tape in three participants was reported.
DiscussionMeta-analyses22-23,25-26,29-31 provided low certainty evidence that exercise interventions compared to no exercise were effective in reducing pain in people with OPVFs. Meta-analyses34,36 provided low certainty evidence that rigid bracing intervention compared with no bracing was effective in reducing pain in patients with OPVFs. No health-related quality of life or activity improvements were demonstrated for exercise interventions, bracing, electrotherapy, or multimodal interventions. Meta-analyses showed low to moderate certainty evidence of no differences in harms comparing exercise and no exercise groups. Our review includes six trials not included in previous reviews and systematically evaluated the effect of non-surgical and non-pharmacological management of OPVFs on harms as well as benefits.
One explanation for why exercise and bracing reduced pain could be both these interventions reduced loading on the spine. A common component of the programs included back extension strengthening or postural control, which reduces flexion and loading on the spine. Takahashi et al.44 found increased load during trunk flexion in healthy adults. Rohlmann et al.45 demonstrated high forces in spinal segments in activities of daily living that involved moving the center of mass anteriorly for example upper body flexion, lifting, or stair walking in patients with instrumented vertebral body replacements. Interestingly, walking was ranked as having the lowest resultant force (N). Pain in people with OPVFs is typically increased during loading activities such as bending and eased when lying down.46
While the results on outcomes for exercise and bracing appear promising for reducing pain, their clinical significance requires further consideration. For example, effect of exercise versus no exercise on pain, (MD = 1.01 units, 95% CI: 0.08, 1.93), is less than the minimum clinically important difference of 1.8 units.47 The pain outcome for bracing (MD = 2.61 units, 95%CI: 0.95, 4.27) exceeds the minimum clinically important difference; however, the lower band of the confidence interval is less than 1.8 units suggesting there remain some uncertainty about the clinical significance of the effect of rigid bracing on pain reduction for people with OPVFs. While there may be small benefits for pain, there was not a change in activity level or quality of life from exercise, bracing, or multimodal interventions. One possible explanation was that the small reduction in pain may not have been large enough to elicit a change in activity level or quality of life.
Even though the effects on pain may be relatively small and the clinical significance questioned, the effects of exercise and bracing compared to no exercise or brace respectively is consistent with management of other back conditions. A recent meta-analysis focused on exercise compared with no treatment/usual care in participants with persistent non-specific low back pain, demonstrated that exercise therapy on average reduced pain by 1.1 units on 0-10 visual analogue scale (95% CI: 0.74, 1.41),48 which is of similar magnitude to our results. Also, these results may be similar to the effects of commonly prescribed medications. One trial demonstrated significantly lower pain intensity in a group receiving tramadol (100 mg/day) compared to a control group allowed to receive a Chinese medicine for pain relief (0.32 g ‘as needed’), at both immediate- (SMD = -1.23, 95% CI: -2.42, -0.05) and short-term follow-ups (SMD = -1.58, 95% CI: -2.83, -0.33).10
The results of this review provide some support for prescribing exercise programs for people with OPVFs as they may have a small effect on pain and avert a small risk of harm. These results add to the uncertain evidence that exercise programs may also have a role in preventing fragility fractures associated with osteoporosis such as OPVFs.49 It might provide support for exercise as a first-line intervention for management of OPVFs before considering common treatments with questionable efficacy and known harms such as analgesia. However, this review has not considered cost effectiveness. Most programs involved supervision which may increase costs and adherence was as low as 58% completion of prescribed sessions. However, our preliminary data suggest the costs of exercise interventions were similar to standard care.41-42 A recent paper by Ferreira et al.,50 found that people with low back pain prefer brief low-cost home-based programs. Perhaps a simple low-cost, less resource-intense intervention for people with OPVFs that retains the important components (e.g. load reduction with relative extension and extensor muscle activation) and therefore may achieve the key benefits may be worthwhile. For example, a walking program might achieve the same benefits and also achieve better adherence. None of the exercise trials in this review included a walking program and this may be an area of further research.
This review followed the PRISMA reporting guidelines for systematic reviews and meta-analyses. All the trials included are randomized controlled trials increasing confidence in results, as findings are less subject to bias. The GRADE approach was applied to meta-analyses determining level of certainty in results.
A limitation of our search strategy was that we used a database filter to limit the search to randomized controlled trials rather than a validated filter such as those used in Cochrane reviews. This may have increased the risk of missing potentially relevant trials. However, the small number of additional trials identified through citation tracking suggests our search strategy was sufficiently sensitive. Only trials reported in English were included in our search, which is a limitation of this review. Computation of results for meta-analyses was immediately post intervention only; longer-term results have not been presented in this review. Related to this, each trial included employed variable length interventions, and this also could be considered a limitation. Finally, the fact that all trials were completed in countries classified as high or upper-middle income status means results may not be generalisable to countries of lower socio-economic status.
ConclusionThere is low certainty evidence that exercise interventions and rigid bracing can reduce pain in people with OPFVs without increasing risk of harm. No benefit for health-related quality of life and activity measures were demonstrated for exercise interventions, bracing, or multimodal interventions. Randomized trials testing the effectiveness of simple low-cost exercise interventions for example walking are needed.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.